O) oxides of caesium are known. Caesium oxide may refer to: Caesium suboxides (Cs7O, Cs4O, and Cs11O3) Caesium monoxide (Cs2O, the most common oxide) Caesium...
1 KB (144 words) - 16:35, 7 December 2023
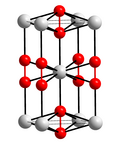
Caesium monoxide or caesium oxide is a chemical compound with the chemical formula Cs2O. It is the simplest and most common oxide of the caesium. It forms...
5 KB (275 words) - 10:51, 14 September 2024
Caesium (IUPAC spelling; also spelled cesium in American English) is a chemical element; it has symbol Cs and atomic number 55. It is a soft, silvery-golden...
93 KB (10,019 words) - 18:02, 23 September 2024
hydroxide: Rb2O + H2O → 2 RbOH Caesium oxide reacts with water to produce caesium hydroxide: Cs2O + H2O → 2 CsOH Magnesium oxide reacts with water to produce...
5 KB (573 words) - 11:18, 30 August 2024
Potassium oxide (K2O) is a pale yellow solid that decomposes at 350 °C. Rubidium oxide (Rb2O) is a yellow solid that melts at 500 °C. Caesium oxide (Cs2O)...
5 KB (457 words) - 17:49, 9 February 2024
Sodium oxide is a chemical compound with the formula Na2O. It is used in ceramics and glasses. It is a white solid but the compound is rarely encountered...
7 KB (542 words) - 05:02, 12 September 2024
Potassium oxide (K2O) is an ionic compound of potassium and oxygen. It is a base. This pale yellow solid is the simplest oxide of potassium. It is a highly...
7 KB (549 words) - 09:43, 22 June 2024
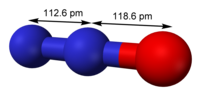
Nitrous oxide (dinitrogen oxide or dinitrogen monoxide), commonly known as laughing gas, nitrous, factitious air, among others, is a chemical compound...
94 KB (9,702 words) - 10:28, 29 September 2024
Lithium oxide (Li 2O) or lithia is an inorganic chemical compound. It is a white solid. Although not specifically important, many materials are assessed...
7 KB (480 words) - 18:59, 8 March 2024
Caesium superoxide is a chemical compound with the chemical formula CsO2. It consists of caesium cations Cs+ and superoxide anions O−2. It is an orange...
3 KB (195 words) - 11:21, 29 December 2023
materials are highly reactive and slurries are useful in synthesis. Caesium oxide is probably the strongest base according to quantum-chemical calculations...
12 KB (1,181 words) - 02:46, 21 November 2023
Caesium hydroxide is a strong base (pKa= 15.76) containing the highly reactive alkali metal caesium, much like the other alkali metal hydroxides such...
6 KB (380 words) - 23:33, 18 March 2024
in a highly dilute solution. Reaction of ethylene oxide with sulfur dioxide in the presence of caesium salts leads to the formation of an 11-membered heterocyclic...
106 KB (11,529 words) - 16:25, 27 September 2024
Caesium selenide is an inorganic compound of caesium and selenium. It is a selenide, with the chemical formula of Cs2Se. It can be prepared by reacting...
3 KB (159 words) - 03:32, 18 June 2023
Caesium sesquioxide is a chemical compound with the formula Cs2O3 or more accurately Cs4O6. It is an oxide of caesium containing oxygen in different oxidation...
8 KB (694 words) - 11:58, 16 July 2024
formaldehyde takes place over a fixed bed of catalyst. This catalyst, caesium oxide on silica, achieves good selectivity to MMA from MeP. The formation...
19 KB (1,711 words) - 20:27, 28 September 2024
of sodium tetrachloroaurate (Na[AuCl4]), caesium carbonate is very efficient mechanism for aerobic oxidation of different kinds of alcohols into ketones...
12 KB (1,084 words) - 13:27, 31 July 2024
decomposition of caesium permanganate leads to the formation of caesium manganate intermediates. It breaks down into manganese dioxide, caesium oxide and oxygen...
4 KB (328 words) - 19:14, 16 September 2024
Alkali metal (section Oxides and chalcogenides)
chemical elements lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute group 1...
214 KB (23,508 words) - 12:55, 26 September 2024
Rubidium oxide is the chemical compound with the formula Rb2O. Rubidium oxide is highly reactive towards water, and therefore it would not be expected...
7 KB (562 words) - 06:53, 12 September 2024
Francium (redirect from Eka-caesium)
22 minutes. It is the second-most electropositive element, behind only caesium, and is the second rarest naturally occurring element (after astatine)...
36 KB (4,059 words) - 01:17, 19 August 2024
Caesium peroxide or cesium peroxide is an inorganic compound of caesium and oxygen with the chemical formula Cs2O2. It can be formed from caesium metal...
4 KB (344 words) - 22:31, 12 April 2024
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to other atoms were fully ionic. It...
45 KB (14,045 words) - 17:32, 4 September 2024
P391, P405, P501 Related compounds Other anions Caesium oxide Caesium selenide Caesium telluride Caesium polonide Other cations Lithium sulfide Sodium sulfide...
4 KB (256 words) - 22:37, 12 April 2024
spellings may differ, such as aluminum/aluminium, sulfur/sulphur, and caesium/cesium. Contents A B C Ca–Cu D E F G H I K L M N O P R S T U V W X Y Z...
183 KB (107 words) - 22:11, 6 September 2024
Cs2CrO4 caesium chromate 56320–90–2 Cs2MoO4 caesium molybdate 13597–64–3 Cs2O caesium oxide 20281–00–9 Cs2O2 caesium peroxide 12053–70–2 Cs2S caesium sulfide...
139 KB (120 words) - 17:07, 15 July 2024
Cuprate (section Oxide cuprates)
CsCuO2 (black) can be prepared by reaction of rubidium oxide and caesium oxide with copper(II) oxide powders, at 675 K (402 °C; 755 °F) and 655 K (382 °C;...
14 KB (1,490 words) - 12:53, 19 September 2024
Caesium chromate is mainly obtained from the reaction of chromium(VI) oxide with caesium carbonate, wherein carbon dioxide gas is evolved: CrO3(aq) + Cs2CO3(aq)...
5 KB (390 words) - 20:50, 12 May 2024
Caesium ozonide is an oxygen-rich chemical compound of caesium, with the chemical formula CsO3. It consists of caesium cations Cs+ and ozonide anions O−3...
4 KB (314 words) - 11:34, 8 December 2023
Caesium lithium borate or cesium lithium borate (CsLiB6O10), also known as CLBO, is a non-linear crystal for ultraviolet applications and generates the...
3 KB (178 words) - 19:40, 12 August 2023