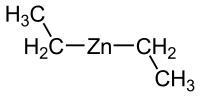Diiodomethane or methylene iodide, commonly abbreviated "MI", is an organoiodine compound. Diiodomethane is a very dense colorless liquid; however, it...
7 KB (412 words) - 14:22, 14 April 2024
potassium tetraiodomercurate(II) (Thoulets solution), bromoform or diiodomethane which are used in mineralogy are very toxic. These toxic chemicals are...
4 KB (323 words) - 16:10, 19 February 2024
a halogen atom from the solvent. For the synthesis of aryl iodides diiodomethane is used, whereas bromoform is the solvent of choice for the synthesis...
19 KB (1,859 words) - 04:31, 25 September 2024
aryl halides. The solvent of choice for the synthesis of iodoarenes is diiodomethane, while for the synthesis of bromoarenes, bromoform is used. For the...
16 KB (1,738 words) - 05:14, 17 August 2024
compound prepared using the Simmons–Smith reaction, by the action of diiodomethane and a zinc-copper couple on cyclohexene in diethyl ether. Smith, R....
2 KB (55 words) - 21:15, 20 July 2021
+ NaOH → CH2Br2 + Na3AsO4 + NaBr Another way is to prepare it from diiodomethane and bromine. Dibromomethane is used as a solvent, gauge fluid, and in...
7 KB (399 words) - 02:57, 10 June 2024
700 Gneiss 2,700 Aluminium 2,700 Limestone 2,750 Compact Basalt 3,000 Diiodomethane 3,325 Liquid at room temperature Diamond 3,500 Titanium 4,540 Selenium...
35 KB (3,297 words) - 17:44, 8 August 2024
alpha-position. Some reagents (e.g. hydrogen iodide) convert iodoform to diiodomethane. Also conversion to carbon dioxide is possible: Iodoform reacts with...
9 KB (738 words) - 13:21, 11 September 2024
the product and the reaction is stereospecific. Thus, cyclohexene, diiodomethane, and a zinc-copper couple (as iodomethylzinc iodide, ICH2ZnI) yield...
27 KB (2,906 words) - 17:06, 26 September 2024
diazomethane (linear CH 2=N 2), diazirine (cyclic [-CH 2-N=N-]) and diiodomethane (I-CH 2-I). The decomposition can be effected by photolysis, photosensitized...
15 KB (1,508 words) - 13:40, 12 September 2024
Simmons–Smith reaction. For example, iodomethylzinc iodide, formed in situ from diiodomethane and a zinc-copper couple reacts with cyclohexene to give norcarane (bicyclo[4...
865 bytes (74 words) - 17:50, 27 October 2019
among the densest fluids known (except some liquid metals), such as diiodomethane. Near 0 °Bé would be approximately the density of water. −100 °Bé (specific...
5 KB (753 words) - 02:59, 13 September 2024
two probe liquids, as described above, and the recommended ones are diiodomethane, which should have no polar component due to its molecular symmetry...
29 KB (4,433 words) - 08:03, 15 August 2024
danger) Ca [100 ppm] Related compounds Related iodomethanes Methane Diiodomethane Iodoform Carbon tetraiodide Except where otherwise noted, data are given...
25 KB (2,559 words) - 10:53, 1 July 2024
small quantities, at the marine boundary layer by the photooxidation of diiodomethane, which is produced by macroalga such as seaweed or through the oxidation...
8 KB (738 words) - 16:42, 4 August 2024
Germanium tetraiodide Tin(IV) iodide Related alkanes Methane Methyl iodide Diiodomethane Iodoform Ethane Ethyl iodide Related compounds Pimagedine Guanidine...
6 KB (446 words) - 06:49, 14 August 2024
m−3 (oral, rat) Related compounds Related iodoalkanes Methyl iodide Diiodomethane Iodoform Carbon tetraiodide n-Propyl iodide Isopropyl iodide Related...
5 KB (227 words) - 23:24, 24 June 2024
of the surface is measured with several liquids, usually water and diiodomethane. Based on the contact angle results and knowing the surface tension...
33 KB (4,049 words) - 19:45, 10 May 2024
can make them easy to identify if unset. High-density liquids such as diiodomethane can be used for this purpose, but these liquids are all highly toxic...
37 KB (5,274 words) - 01:55, 26 September 2024
in the reaction of samarium metal with either diiodomethane or diiodoethane. The use of diiodomethane is convenient because the oxidant is a liquid (diiodoethane...
17 KB (1,966 words) - 20:27, 29 August 2024
prepared in nearly quantitative yields from samarium metal and either diiodomethane or 1,2-diiodoethane. When prepared in this way, its solutions is most...
11 KB (958 words) - 20:35, 17 September 2024
as a reagent for cyclopropanation by the Simmons-Smith reaction, but diiodomethane and chloroiodomethane are preferred. It also occurs naturally as the...
5 KB (306 words) - 18:34, 11 September 2024
in cyclopropanation (Simmon-Smith reaction), where it often replaces diiodomethane because of higher yields and selectivity. It is also used in Mannich...
4 KB (224 words) - 05:41, 28 March 2023
benzaldehyde and imines. Additionally, it is commonly used in combination with diiodomethane as a Simmons-Smith reagent to convert alkenes into cyclopropyl groups...
12 KB (1,156 words) - 22:49, 26 August 2024
iodomethylzinc iodide, which is typically formed by a reaction between diiodomethane and a zinc-copper couple. Modifications involving cheaper alternatives...
14 KB (1,504 words) - 19:07, 5 July 2024
carbene is added to the double bond of the unsaturated fatty acid; diiodomethane and zinc are used to form the carbene. The Simmons-Smith reaction is...
39 KB (4,480 words) - 09:56, 15 September 2024
Oliver Chenpu Ho (April 1990). "Application of the isoamyl nitrite-diiodomethane route to aryl iodides". The Journal of Organic Chemistry. 55 (8): 2543–2545...
3 KB (165 words) - 22:24, 8 January 2024
halogen atom: difluoromethane, dichloromethane, dibromomethane and diiodomethane. There are six members with two kinds of halogen atoms: Bromochloromethane...
2 KB (157 words) - 18:53, 11 May 2024
Simmons-Smith cyclopropanation, which employs carbenes derived from diiodomethane, is a popular alternative to rhodium-catalyzed cyclopropanation. In...
11 KB (1,251 words) - 20:49, 22 July 2024
4.21 1.00 1.16 0.83 3-methylhexane 146.4 14.95 0.00 1.00 0.00 0.00 diiodomethane 81.0 21.90 5.19 1.00 2.40 2.08 2,2-dimethylpentane 148.9 14.26 0.00...
26 KB (1,482 words) - 23:33, 19 July 2022