Disilane is a chemical compound with chemical formula Si2H6 that was identified in 1902 by Henri Moissan and Samuel Smiles (1877–1953). Moissan and Smiles...
5 KB (452 words) - 04:32, 31 May 2024
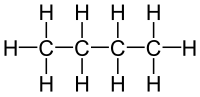
starting with silane, SiH4, the analogue of methane, continuing with disilane Si2H6, the analogue of ethane, etc. They are mainly of theoretical or academic...
9 KB (967 words) - 16:55, 24 May 2024
experimentally. Silicon can form sigma bonds to other silicon atoms (and disilane is the parent of this class of compounds). However, it is difficult to...
12 KB (1,465 words) - 04:12, 15 April 2024
Alvarez, S (2002). "Hexakis(silyl)palladium(VI) or palladium(II with eta2-disilane ligands?". Angewandte Chemie International Edition in English. 41 (11):...
59 KB (6,283 words) - 17:25, 19 August 2024
'tricarbane', and 'tetracarbane' as recommended for analogues of silane, 'disilane'; phosphane, 'triphosphane'; and sulfane, 'tetrasulfane'. W. B. Kay (1940)...
23 KB (2,095 words) - 18:56, 25 August 2024
'tricarbane', and 'tetracarbane' as recommended for analogues of silane, 'disilane'; phosphane, 'triphosphane'; and sulfane, 'tetrasulfane'." "Ethane – Compound...
30 KB (3,021 words) - 10:07, 11 August 2024
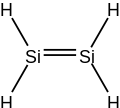
5 to 10% shorter than the Si-Si single bond lengths of corresponding disilanes. A peculiarity of disilenes is the trans-bending of the substituents,...
5 KB (568 words) - 02:22, 22 October 2023
also) includes silane itself but also compounds with Si-Si bonds including disilane and longer chains. Silanes with one, two, three, or four Si-H bonds are...
3 KB (308 words) - 07:20, 2 June 2024
Methane Germane Stannane Plumbane Related compounds Phenylsilane Vinylsilane Disilane Trisilane Except where otherwise noted, data are given for materials in...
20 KB (2,204 words) - 01:47, 4 September 2024
'tricarbane', and 'tetracarbane' as recommended for analogues of silane, 'disilane'; phosphane, 'triphosphane'; and sulfane, 'tetrasulfane'. Lide, David R...
50 KB (5,441 words) - 20:20, 4 September 2024
sodium fluoride: 6 NaH + 2 BF3 → B2H6 + 6 NaF Si–Si and S–S bonds in disilanes and disulfides are also reduced. A series of reduction reactions, including...
14 KB (1,339 words) - 02:55, 29 March 2024
which thermal stability decreasing as n increases (e.g. silane SiH4 and disilane Si2H6), as are cyclosilanes (e.g. cyclopentasilane Si5H10 and cyclohexasilane...
9 KB (818 words) - 15:44, 25 June 2023
NO2Cl −15 −145 81.5 13444-90-1 Chlorazide ClN3 −15 −100 77.47 13973-88-1 Disilane Si2H6 −14.8 −129.4 62 1590-87-0 Z-1,2,3,3,3-Pentafluoropropene CHF=CFCF3...
95 KB (4,549 words) - 10:45, 10 August 2024
silane) (should not be called siliconium disilanium, Si2H+7 (protonated disilane) further silanium cations, Si nH+ 2n+3 (protonated silanes) germonium,...
16 KB (1,499 words) - 15:22, 4 December 2023
that a B=B double bond is occurring. With specific reaction conditions, a disilane single or double bond can be achieved using the NHC boryl anion. To make...
22 KB (2,511 words) - 05:04, 18 June 2024
that link the silicene hexagons use disilyne bonding alternating with disilane bonding, analogous to polyyne. Phillip F. Schewe & Ben Stein (3 September...
3 KB (314 words) - 18:04, 23 August 2024
thiocyanate – CNSCN Cyanogen selenocyanate – CNSeCN Cyanogen azide – N3CN Disilane – Si2H6 Disulfur dichloride – S2Cl2 Dysprosium(III) chloride – DyCl3 Dysprosium...
119 KB (8,729 words) - 14:41, 31 August 2024
for terminal alkenes. Aside from boranes, the oxidation of silanes and disilanes can also yield hydroxy groups. A major difference is that while silyl...
8 KB (942 words) - 15:12, 8 July 2024
structure, sometimes more are produced from the same compound; e.g. silane, disilane and trisilane from decomposition of magnesium silicide) Germanes from germanides...
11 KB (1,332 words) - 18:54, 17 April 2023
3-bis(2,6-diisopropylphenyl)imidazolium fluoride. In combination with disilane, germanium tetrafluoride is used for in the synthesis of SiGe. Germanium(IV)...
5 KB (298 words) - 22:00, 27 March 2024
polymerizes. As it is heated more silane is produced, and at 178°C it yields disilane. When heated to over 250°C it decomposes to a silicon hydrogen polymer...
4 KB (393 words) - 00:18, 23 May 2023
(fire diamond) 2 4 1 Related compounds Related compounds Dimethyl ether Disilane Silane Silanol Trisilane Except where otherwise noted, data are given for...
9 KB (895 words) - 06:19, 23 January 2024
magnesium silicide with 25% hydrochloric acid produces 40% monosilane, 30% disilane, 15% trisilane, 10% tetrasilane and 5% higher silanes. The mixture can...
5 KB (612 words) - 07:27, 27 August 2024
as SiH 4 and SiHCl 3 by Charles Friedel and Albert Ladenburg in 1867. Disilane (Si 2H 6) followed in 1902, when it was first made by Henri Moissan and...
42 KB (5,607 words) - 22:19, 30 May 2024
110 °C in xylene thereby releasing its strain energy[clarification needed]. Disilane Disilyne McCarthy, M. C.; Yu, Z.; Sari, L.; Schaefer, H. F.; Thaddeus,...
4 KB (375 words) - 19:07, 19 January 2024
extended compared to the gas phase Si-Si bond length of 2.331Å in the parent disilane H3SiSiH3. Despite the long bond length, superdisilane exhibits thermal...
24 KB (2,637 words) - 15:21, 22 August 2024
Fürstner, Alois; Weidmann, Hans (1988). "Efficient formation and cleavage of disilanes by potassium-graphite. Silylation with silyl metal reagents". Journal...
4 KB (181 words) - 19:50, 1 January 2023
batteries packed with equipment, with organic electrolyte UN 3553 2.1 Disilane UN 3554 8 Gallium contained in manufactured articles UN 3555 3...
7 KB (115 words) - 23:34, 5 August 2024
monosilane (SiH4) and disilane (Si2H6). Polysilicon hydrides are intermediates in the high-temperature conversion of mono- and disilane to silicon and hydrogen...
8 KB (1,005 words) - 18:37, 26 October 2023
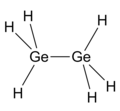
pyrolysis has been found to be more endothermic than the pyrolysis of disilane. This difference is attributed to the greater strength of the Ge-H bond...
8 KB (779 words) - 01:26, 25 May 2023