Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target,...
59 KB (4,170 words) - 01:29, 13 October 2024
A monoclonal antibody (mAb, more rarely called moAb) is an antibody produced from a cell lineage made by cloning a unique white blood cell. All subsequent...
48 KB (4,964 words) - 23:05, 30 September 2024
nomenclature of monoclonal antibodies is a naming scheme for assigning generic, or nonproprietary, names to monoclonal antibodies. An antibody is a protein...
38 KB (3,088 words) - 05:02, 8 August 2024
usage. See the list of FDA-approved therapeutic monoclonal antibodies in the monoclonal antibody therapy page. World Health Organization (2007). "International...
136 KB (4,079 words) - 00:12, 12 October 2024
"humanization" is usually applied to monoclonal antibodies developed for administration to humans (for example, antibodies developed as anti-cancer drugs)...
14 KB (1,787 words) - 23:05, 9 January 2024
Antibody therapy may refer to several different uses of antibodies for the treatment of medical conditions. Monoclonal antibody therapy Oligoclonal antibody...
294 bytes (61 words) - 19:16, 14 February 2023
controlled by interruption of the complement cascade. Particular monoclonal antibodies, discussed later in the article, have proven efficacy in many cases...
38 KB (4,164 words) - 20:08, 15 October 2024
A bispecific monoclonal antibody (BsMAb, BsAb) is an artificial protein that can simultaneously bind to two different types of antigen or two different...
30 KB (3,405 words) - 20:39, 11 October 2024
population studies., Antibody allotypes came back to spotlight due to development and use of therapies based on monoclonal antibodies. These recombinant...
9 KB (1,037 words) - 05:53, 19 August 2023
Otilimab (category Drugs that are a monoclonal antibody)
HuCAL standing for Human Combinatorial Antibody Library and being a technology used to generate monoclonal antibodies. Otilimab is directed against the...
8 KB (722 words) - 15:17, 22 March 2023
Omalizumab (redirect from Monoclonal anti-IgE antibodies)
allergy. Omalizumab is a recombinant DNA-derived humanized IgG1 monoclonal antibody which specifically binds to free human immunoglobulin E (IgE) in...
47 KB (4,925 words) - 23:04, 16 August 2024
Muromonab-CD3 (redirect from Muromonab-CD3 monoclonal antibody)
for the therapy of acute, glucocorticoid-resistant rejection of allogeneic kidney, heart, and liver transplants. Unlike the monoclonal antibodies basiliximab...
13 KB (1,169 words) - 19:22, 17 July 2024
(18F) labeling of antibodies, which allows for positron emission tomography (PET) imaging of cancer. Targeted monoclonal antibody therapy is employed to...
119 KB (13,400 words) - 17:29, 29 August 2024
Rituximab (category Drugs that are a monoclonal antibody)
Rituximab, sold under the brand name Rituxan among others, is a monoclonal antibody medication used to treat certain autoimmune diseases and types of...
62 KB (5,640 words) - 05:11, 1 October 2024
chaperone therapy molecular therapy monoclonal antibody therapy monotherapy mud therapy (prescientific) music therapy negative air ionization therapy neoadjuvant...
8 KB (649 words) - 02:40, 23 September 2024
Cancer immunotherapy (redirect from Checkpoint antibody)
Naked monoclonal antibodies are antibodies without added elements. Most antibody therapies use this antibody type. Conjugated monoclonal antibodies are...
92 KB (12,517 words) - 05:38, 1 October 2024
Immunoconjugate (redirect from Antibody-conjugated toxin)
needed] and to develop monoclonal antibody therapy as a targeted form of chemotherapy when they are often known as antibody-drug conjugates. When the...
2 KB (155 words) - 04:46, 13 August 2023
Lokivetmab (category Drugs that are a monoclonal antibody)
was the first monoclonal antibody to be approved for use in animals in the European Union. "Zoetis earns license for Cytopoint skin therapy". Veterinary...
3 KB (215 words) - 23:23, 12 June 2024
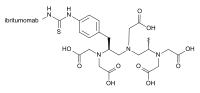
Ibritumomab tiuxetan (category Drugs that are a monoclonal antibody)
Zevalin, is a monoclonal antibody radioimmunotherapy treatment for non-Hodgkin's lymphoma. The drug uses the monoclonal mouse IgG1 antibody ibritumomab...
9 KB (746 words) - 05:56, 25 July 2024
COVID-19 antibody drug was paused due to lack of efficacy against the emerging omicron variant. A second COVID-19 monoclonal antibody therapy, bebtelovimab...
165 KB (15,040 words) - 17:42, 15 October 2024
Bamlanivimab (category Drugs that are a monoclonal antibody)
Bamlanivimab is a monoclonal antibody developed by AbCellera Biologics and Eli Lilly as a treatment for COVID-19. The medication was granted an emergency...
19 KB (1,642 words) - 04:38, 24 July 2024
Passive antibody therapy, also called serum therapy, is a subtype of passive immunotherapy that administers antibodies (same as immunoglobin) to target...
27 KB (2,923 words) - 15:03, 4 April 2024
examples of monoclonal antibodies. For several decades, and until recently, mice were used extensively in the production of monoclonal antibodies (MAbs)....
7 KB (822 words) - 18:53, 4 May 2024
Biopharmaceutical (redirect from Biologic therapy)
the remainder of life. The cost of treatment with a typical monoclonal antibody therapy for relatively common indications is generally in the range of...
30 KB (2,882 words) - 11:11, 23 July 2024
Casirivimab/imdevimab (category Drugs that are a monoclonal antibody)
treatment and prevention of COVID‑19. It consists of two human monoclonal antibodies, casirivimab and imdevimab that must be mixed together and administered...
49 KB (3,417 words) - 23:48, 14 October 2024
Antiserum (redirect from Serum therapy)
In immunology, antiserum is a blood serum containing antibodies (either monoclonal or polyclonal) that is used to spread passive immunity to many diseases...
20 KB (2,374 words) - 01:40, 6 September 2024
antiserum therapy by immunoglobulins: immunoglobulin therapy by monoclonal antibodies: monoclonal antibody therapy by urine: urine therapy (some scientific...
29 KB (2,492 words) - 08:20, 6 August 2024
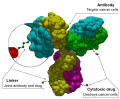
Antibody–drug conjugates or ADCs are a class of biopharmaceutical drugs designed as a targeted therapy for treating cancer. Unlike chemotherapy, ADCs are...
41 KB (4,305 words) - 11:56, 24 August 2024
COVID-19 (section Passive antibodies)
issued an emergency use authorisation for the investigational monoclonal antibody therapy bamlanivimab for the treatment of mild-to-moderate COVID‑19....
288 KB (33,229 words) - 20:37, 10 October 2024
to monoclonal antibodies produced by hybridoma technology, which may lose the capacity to produce the desired antibody over time or the antibody may...
28 KB (3,348 words) - 12:40, 6 August 2024