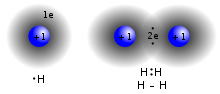
In chemistry, a hydrogen bond (or H-bond) is primarily an electrostatic force of attraction between a hydrogen (H) atom which is covalently bonded to a...
46 KB (5,422 words) - 04:08, 12 August 2024
the carbon-hydrogen bond (C−H bond) is a chemical bond between carbon and hydrogen atoms that can be found in many organic compounds. This bond is a covalent...
6 KB (534 words) - 15:22, 31 January 2024
hydrogen bond is a special type of hydrogen bond in which the proton is spaced exactly halfway between two identical atoms. The strength of the bond to...
2 KB (184 words) - 23:31, 9 March 2020
Intermolecular force (redirect from Dipole-dipole bond)
categorized into the following types: Hydrogen bonding Ion–dipole forces and ion–induced dipole force Cation–π, σ–π and π–π bonding Van der Waals forces – Keesom...
28 KB (3,469 words) - 10:10, 5 August 2024
such as dipole–dipole interactions, the London dispersion force, and hydrogen bonding. Since opposite electric charges attract, the negatively charged electrons...
40 KB (4,876 words) - 20:13, 24 May 2024
structures by non-covalent hydrogen bonding interactions. The directionality, reversibility, and strong bonding nature of hydrogen bond make it an attractive...
12 KB (1,308 words) - 01:11, 14 May 2024
"co-valent bond", in essence, means that the atoms share "valence", such as is discussed in valence bond theory. In the molecule H 2, the hydrogen atoms share...
28 KB (3,654 words) - 19:53, 20 August 2024
Hydrogen-bond catalysis is a type of organocatalysis that relies on use of hydrogen bonding interactions to accelerate and control organic reactions....
32 KB (3,988 words) - 02:45, 1 July 2024
to strong and extensive hydrogen bonding, it boils at near room temperature, much higher than other hydrogen halides. Hydrogen fluoride is an extremely...
15 KB (1,373 words) - 04:58, 14 August 2024
oxidizer in rocketry. Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes...
91 KB (9,212 words) - 18:28, 20 August 2024
carbon–hydrogen bond activation (C−H activation) is a type of organic reaction in which a carbon–hydrogen bond is cleaved and replaced with a C−X bond (X...
36 KB (3,912 words) - 15:15, 17 August 2024
Amide (redirect from Amide bond)
groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs...
21 KB (2,073 words) - 22:46, 27 July 2024
molecule, the bond-dissociation energy of an oxygen–hydrogen bond varies slightly depending on whether or not there is another hydrogen atom bonded to...
9 KB (1,318 words) - 02:44, 29 April 2024
Properties of water (redirect from Hydrogen Hydroxide)
dissociate ions in salts and bond to other polar substances such as alcohols and acids, thus dissolving them. Its hydrogen bonding causes its many unique properties...
88 KB (9,550 words) - 13:45, 9 August 2024
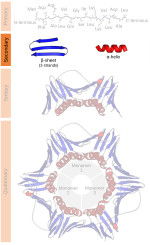
Beta sheet (section Hydrogen bonding patterns)
proposed by William Astbury in the 1930s. He proposed the idea of hydrogen bonding between the peptide bonds of parallel or antiparallel extended β-strands...
27 KB (3,120 words) - 01:54, 14 August 2024
Non-canonical base pairs are planar hydrogen bonded pairs of nucleobases, having hydrogen bonding patterns which differ from the patterns observed in...
89 KB (11,719 words) - 14:07, 29 July 2024
mechanics through the exploration of its energetics and chemical bonding. Hydrogen gas was first produced artificially in the early 16th century by reacting...
121 KB (12,379 words) - 18:04, 21 August 2024
Non-covalent interaction (redirect from Non-covalent bond)
(Na+). A hydrogen bond (H-bond), is a specific type of interaction that involves dipole–dipole attraction between a partially positive hydrogen atom and...
28 KB (3,363 words) - 00:54, 15 December 2023
Beta turn (section Hydrogen bond criterion)
can be defined in two ways: By the possession of an intra-main-chain hydrogen bond between the CO of residue i and the NH of residue i+3; By having a distance...
9 KB (1,100 words) - 03:01, 2 March 2024
Atoms in molecules (redirect from Hydrogen hydrogen bond)
two ortho hydrogen atoms again is shorter than their van der Waals radii and according to in silico experiments based on this theory, a bond path is identified...
12 KB (1,525 words) - 10:35, 20 August 2024
Chemical polarity (redirect from Polar bond)
polarity if the bond dipoles cancel each other out by symmetry. Polar molecules interact through dipole-dipole intermolecular forces and hydrogen bonds. Polarity...
24 KB (2,751 words) - 06:38, 15 August 2024
formula HCl. Hydrogen chloride is a diatomic molecule, consisting of a hydrogen atom H and a chlorine atom Cl connected by a polar covalent bond. The chlorine...
27 KB (2,805 words) - 05:57, 13 August 2024
Salicylaldehyde (section Internal hydrogen bonding)
internal hydrogen bond is formed between the groups. The hydroxy group serves here as the hydrogen bond donor, and the aldehyde as hydrogen bond acceptor...
9 KB (774 words) - 06:21, 21 August 2024
Molecular solid (section Hydrogen and halogen bonding)
interactions, quadrupole interactions, π–π interactions, hydrogen bonding, halogen bonding, London dispersion forces, and in some molecular solids, coulombic...
33 KB (2,913 words) - 15:36, 1 April 2024
placing the peptide group in a hydrophobic environment or donating a hydrogen bond to the nitrogen atom of an X-Pro peptide group. Both of these mechanisms...
12 KB (1,388 words) - 05:15, 27 April 2024
Alpha helix (section Geometry and hydrogen bonding)
with 13 atoms being involved in the ring formed by the hydrogen bond (starting with amidic hydrogen and ending with carbonyl oxygen) In the early 1930s,...
46 KB (5,219 words) - 01:46, 14 August 2024
Base pair (section Hydrogen bonding and stability)
to the folded structure of both DNA and RNA. Dictated by specific hydrogen bonding patterns, "Watson–Crick" (or "Watson–Crick–Franklin") base pairs (guanine–cytosine...
32 KB (3,677 words) - 09:22, 6 August 2024
helix and π helix, are calculated to have energetically favorable hydrogen-bonding patterns but are rarely observed in natural proteins except at the...
28 KB (3,072 words) - 15:24, 4 February 2024
in hydrogen bonding. These molecules compete with surrounding hydrogen bond acceptors for hydrogen bond donors, therefore acting as "hydrogen bond breakers"...
30 KB (3,178 words) - 21:51, 1 August 2024
A Low-barrier hydrogen bond (LBHB) is a special type of hydrogen bond. LBHBs can occur when the pKa of the two heteroatoms are closely matched, which...
6 KB (687 words) - 20:42, 22 July 2024