Ammonium fluoride is the inorganic compound with the formula NH4F. It crystallizes as small colourless prisms, having a sharp saline taste, and is highly...
6 KB (455 words) - 14:03, 18 September 2024
Ammonium bifluoride is an inorganic compound with the formula [NH4][HF2] or [NH4]F·HF. It is produced from ammonia and hydrogen fluoride. This colourless...
10 KB (776 words) - 15:27, 12 October 2024
known as diammine silver fluoride, silver fluoride, and silver ammonium fluoride. It is frequently spelled "silver diamine fluoride" (with one m); however...
33 KB (3,848 words) - 06:53, 19 May 2024
quaternary ammonium salts of naked fluoride include tetramethylammonium fluoride and tetrabutylammonium fluoride. Cobaltocenium fluoride is another example...
47 KB (5,060 words) - 17:43, 5 October 2024
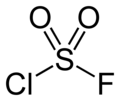
chloride fluoride can be prepared without using gases as starting materials by treating sulfuryl chloride with ammonium fluoride or potassium fluoride in trifluoroacetic...
5 KB (273 words) - 18:15, 2 September 2023
Ammonium chloride is an inorganic chemical compound with the chemical formula NH4Cl, also written as [NH4]Cl. It is an ammonium salt of hydrogen chloride...
47 KB (5,198 words) - 13:05, 26 September 2024
Ammonium bromide, NH4Br, is the ammonium salt of hydrobromic acid. The chemical crystallizes in colorless prisms, possessing a saline taste; it sublimes...
4 KB (215 words) - 17:00, 13 August 2023
in microfabrication. It is a mixture of a buffering agent, such as ammonium fluoride NH4F, and hydrofluoric acid (HF). Its primary use is in etching thin...
5 KB (563 words) - 15:48, 19 September 2024
Tetra-n-butylammonium fluoride, commonly abbreviated to TBAF and n-Bu4NF, is a quaternary ammonium salt with the chemical formula (CH3CH2CH2CH2)4N+F−....
6 KB (492 words) - 02:29, 27 June 2022
fluoride exposure are due to the ingestion of dental fluoride products. Other sources include glass-etching or chrome-cleaning agents like ammonium bifluoride...
25 KB (2,596 words) - 16:35, 11 October 2024
readily produced by the electrolysis of molten ammonium fluoride dissolved in anhydrous hydrogen fluoride. Like carbon tetrafluoride, it is not at all reactive...
105 KB (12,228 words) - 16:47, 4 October 2024
Beryllium fluoride is the inorganic compound with the formula BeF2. This white solid is the principal precursor for the manufacture of beryllium metal...
11 KB (935 words) - 22:08, 26 September 2024
Holmium(III) fluoride is a yellowish powder that can be produced by reacting holmium(III) oxide and ammonium fluoride, then crystallising it from the ammonium salt...
36 KB (4,143 words) - 22:51, 11 September 2024
since it avoids producing toxic waste products such as ammonium fluoride. Copper(II) fluoride can be synthesized from copper and fluorine at temperatures...
11 KB (951 words) - 00:33, 25 September 2024
mineral sellaite. Magnesium fluoride is prepared from magnesium oxide with sources of hydrogen fluoride such as ammonium bifluoride, by the breakdown...
9 KB (564 words) - 21:54, 21 July 2024
Ammonia – NH3 Ammonium azide – [NH4]N3 Ammonium bicarbonate – [NH4]HCO3 Ammonium bisulfate – [NH4]HSO4 Ammonium bromide – NH4Br Ammonium chromate – [NH4]2CrO4...
119 KB (8,735 words) - 14:26, 16 September 2024
heated to decomposition, ammonium tetrafluoroborate releases toxic fumes of hydrogen fluoride, nitrogen oxides, and ammonia. Ammonium tetrafluoroborate can...
5 KB (146 words) - 14:31, 4 September 2023
reaction of gallium(III) hydroxide, HF, and ammonium fluoride. Ga(OH)3 + 3 HF + 3 NH4F → (NH)3GaF6 + 3 H2O Ammonium hexafluorogallate occurs in two polymorphic...
4 KB (297 words) - 07:22, 18 September 2024
hydrogen fluoride. It has also been used as a precursor to zeolites. Ammonium hexafluoroaluminate can be obtained by the reaction of ammonium fluoride and...
3 KB (216 words) - 07:40, 22 August 2024
Holmium(III) fluoride is an inorganic compound with a chemical formula of HoF3. Holmium(III) fluoride can be produced by reacting holmium oxide and ammonium fluoride...
3 KB (237 words) - 15:05, 26 December 2023
acquired the 17 acre site in 2021. Sodium fluoride Hydrofluoric acid Ammonium fluoride Phosphoric acid Hydrogen fluoride Wiles, Russ. "Massive semiconductor...
3 KB (186 words) - 00:21, 26 June 2024
Nitrogen trifluoride (redirect from Nitrogen(III) fluoride)
trifluoride by the electrolysis of a molten mixture of ammonium fluoride and hydrogen fluoride. It is far less reactive than the other nitrogen trihalides...
22 KB (1,964 words) - 06:53, 26 September 2024
+ CO2 Another possible method is to react rubidium hydroxide with ammonium fluoride: RbOH + NH4F → RbF + H2O + NH3 The least used method due to expense...
7 KB (490 words) - 17:35, 26 December 2023
Aluminium fluoride is an inorganic compound with the formula AlF3. It forms hydrates AlF3·xH2O. Anhydrous AlF3 and its hydrates are all colorless solids...
14 KB (1,283 words) - 09:37, 11 April 2024
infrared-absorbing glass. Erbium(III) fluoride is a pinkish powder that can be produced by reacting erbium(III) nitrate and ammonium fluoride. It can be used to make...
34 KB (4,095 words) - 04:24, 28 September 2024
organyl group. Unlike the ammonium ion (NH+4) and the primary, secondary, or tertiary ammonium cations, the quaternary ammonium cations are permanently...
27 KB (2,783 words) - 04:39, 18 September 2024
anion, a weakly coordinating anion. It is prepared by combining neat ammonium fluoride and phosphorus pentachloride. Alternatively it can also be produced...
2 KB (107 words) - 21:36, 10 March 2022
Ammonium iodide is the inorganic compound with the formula NH4I. A white solid. It is an ionic compound, although impure samples appear yellow. This salt...
5 KB (358 words) - 17:42, 12 August 2024
Fluorine (section Fluoride ion)
silicon tetrafluoride. Other important inorganic fluorides include those of cobalt, nickel, and ammonium. Organofluorides consume over 20% of mined fluorite...
157 KB (15,346 words) - 01:04, 8 September 2024
China. Barium fluoride can be prepared by precipitating barium chloride and ammonium fluoride in a solution. Barium chloride fluoride forms white crystals...
4 KB (303 words) - 17:43, 29 April 2024