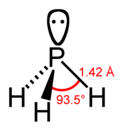
Phosphonium iodide is a chemical compound with the formula PH 4I. It is an example of a salt containing an unsubstituted phosphonium cation (PH+ 4). Phosphonium...
6 KB (531 words) - 03:39, 3 May 2023
take the color of the anions. The parent phosphonium is PH+ 4 as found in the iodide salt, phosphonium iodide. Salts of the parent PH+ 4 are rarely encountered...
13 KB (1,231 words) - 23:45, 1 June 2023
Diphosphorus tetraiodide (redirect from Phosphorus(II) iodide)
phosphorus trichloride and potassium iodide in anhydrous conditions. Another synthesis route involves combining phosphonium iodide with iodine in a solution of...
6 KB (556 words) - 15:37, 1 June 2023
Kohlenwasserstoffe durch Jodphosphonium" [On the reduction of aromatic compound by phosphonium iodide [H4IP]]. Annalen der Chemie und Pharmacie. 55: 266–281. Bei der Reduction...
18 KB (1,559 words) - 10:45, 19 April 2024
Bergmann which demonstrated that with a mixture of hydrogen iodide and phosphonium iodide, p-toluenesulfonyl amino acids could be detosylated reductively...
25 KB (3,025 words) - 11:23, 11 February 2024
Ammonium iodide is the inorganic compound with the formula NH4I. A white solid. It is an ionic compound, although impure samples appear yellow. This salt...
5 KB (358 words) - 17:42, 12 August 2024
Wittig reaction (redirect from Phosphonium ylide)
olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide called a Wittig reagent. Wittig reactions are most commonly used...
15 KB (1,600 words) - 17:50, 27 May 2024
P2H4, may be prepared using the action of potassium hydroxide on phosphonium iodide: [PH4]I + KOH → PH3 + KI + H2O Phosphine is a worldwide constituent...
39 KB (3,593 words) - 22:09, 15 August 2024
Kohlenwasserstoffe durch Jodphosphonium" [On the reduction of aromatic compound by phosphonium iodide [H4IP]]. Annalen der Chemie und Pharmacie. 55: 266–281. Bei der Reduction...
6 KB (557 words) - 01:22, 25 July 2023
Wittig reagents (section Formation of phosphonium salt)
to be sold commercially. Wittig reagents are usually prepared from a phosphonium salt, which is in turn prepared by the quaternization of triphenylphosphine...
7 KB (714 words) - 02:26, 19 June 2024
the acid and base. Phosphonium cations (R4P+) are less common but offer some advantageous properties. Some examples of phosphonium cations are...
43 KB (4,706 words) - 07:07, 9 August 2024
organophosphorus compounds. Compounds with the formula [PR4+]X− comprise the phosphonium salts. These species are tetrahedral phosphorus(V) compounds. From the...
16 KB (1,730 words) - 17:17, 3 June 2024
N-carbobenzyloxy-α-N-carboxy-L-lysine anhydride, followed by deprotection with phosphonium iodide. Peptide synthesis from NCAs does not require protection of the amino...
11 KB (1,112 words) - 15:16, 20 February 2023
Sulfonyl halide (section Sulfonyl iodides)
sulfonyl halides decreases in the order fluorides > chlorides > bromides > iodides, all four types being well known. The sulfonyl chlorides and fluorides...
12 KB (1,352 words) - 19:02, 13 March 2024
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
5 KB (539 words) - 17:56, 16 July 2024
alkyl bromides, whereas using carbon tetraiodide, methyl iodide or iodine gives alkyl iodides. The reaction is credited to and named after Rolf Appel,...
7 KB (685 words) - 00:12, 10 March 2024
that reacts by the SN2 pathway: CH3OH + H+ → [CH3OH2]+ Similarly, methyl iodide and methyl triflate are viewed as the equivalent of the methyl cation because...
12 KB (1,409 words) - 12:36, 8 August 2024
vinyl iodide/phosphonium salt. phosphonium salt could be readily obtained as the trisubstituted vinyl iodide is less reactive than alkyl iodide. A Wittig...
30 KB (3,454 words) - 21:37, 22 May 2024
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
3 KB (261 words) - 01:37, 27 August 2023
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
9 KB (1,044 words) - 16:54, 8 May 2024
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
9 KB (1,122 words) - 09:22, 18 August 2024
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
23 KB (2,633 words) - 07:34, 30 May 2024
(1949-05-10). "Über die Metallierbarkeit von quaternären Ammonium- und Phosphonium-Salzen". Justus Liebigs Annalen der Chemie. 562 (3): 177–186. doi:10...
7 KB (650 words) - 09:15, 19 February 2024
ammonium (IUPAC name azanium), NH+4 (protonated ammonia (IUPAC name azane)) phosphonium, PH+4 (protonated phosphine) arsonium, AsH+4 (protonated arsine) stibonium...
16 KB (1,499 words) - 15:22, 4 December 2023
volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting...
20 KB (2,413 words) - 13:06, 14 April 2024
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
11 KB (1,085 words) - 14:51, 4 July 2024
limits reaction rate. In Simmons-Smith cyclopropanation, the iodomethylzinc iodide typically complexes to any allylic hydroxy groups such that addition is...
22 KB (2,401 words) - 05:50, 13 August 2024
Phosphonate Phosphite Phosphonous Phosphinate Phosphine oxide Phosphine Phosphonium Phosphaalkene Phosphaalkyne Phosphaallene Sulfur Thiol Sulfide Sulfonium...
15 KB (1,654 words) - 00:59, 6 July 2024
nucleophilic toward hard electrophiles. With methyl iodide it forms trimethylsulfoxonium iodide, [(CH3)3SO]I: (CH3)2SO + CH3I → [(CH3)3SO]I This salt...
38 KB (3,969 words) - 09:03, 30 May 2024
readily alkylate to stable sulfonium salts, such as trimethylsulfonium iodide: S(CH3)2 + CH3I → [S(CH3)3]+I− Sulfides also oxidize easily to sulfoxides...
10 KB (1,120 words) - 00:46, 27 April 2024