In chemistry, disulfate or pyrosulfate is the anion with the molecular formula S 2O2− 7. Disulfate is the IUPAC name. It has a dichromate-like structure...
2 KB (248 words) - 17:19, 5 May 2024
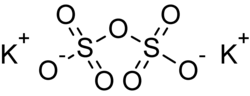
Potassium pyrosulfate, or potassium disulfate, is an inorganic compound with the chemical formula K2S2O7. Potassium pyrosulfate is obtained by the thermal...
6 KB (379 words) - 19:18, 12 June 2023
Sodium pyrosulfate is an inorganic compound with the chemical formula of Na2S2O7. It is a colorless salt. It hydrolyses in water to form sodium bisulfate...
4 KB (257 words) - 21:26, 2 August 2024
FSO3H SO3 reacts with dinitrogen pentoxide to give the nitronium salt of pyrosulfate: 2 SO3 + N2O5 → [NO2]2S2O7 Sulfur trioxide is an oxidant. It oxidizes...
18 KB (1,554 words) - 16:37, 9 October 2024
of potassium bisulfate forms potassium pyrosulfate: 2 KHSO4 → K2S2O7 + H2O Above 600 °C potassium pyrosulfate converts to potassium sulfate and sulfur...
6 KB (330 words) - 13:50, 5 April 2024
is 1.77 Å distant from the two Mn atoms. The Mn−O−Mn angle is 120.7°. Pyrosulfate, pyrophosphate, and dichromate adopt structures similar to that of Mn2O7...
7 KB (535 words) - 08:05, 1 October 2024
exothermically at 120 °C with evolution of oxygen and the formation of the pyrosulfate. AgF2 + H2SO4 → AgSO4 + 2 HF 4 AgSO4 → 2 Ag2S2O7 + O2 Lide, David R....
7 KB (359 words) - 18:48, 10 April 2024
hygroscopic. Heating sodium bisulfate to 280 °C (536 °F) produces sodium pyrosulfate, another colorless salt: 2 NaHSO4 → Na2S2O7 + H2O Sodium bisulfate is...
12 KB (1,044 words) - 05:54, 17 September 2024
Disulfuric acid (category Pyrosulfates)
system. There are salts of disulfuric acid, commonly called pyrosulfates, e.g. potassium pyrosulfate. There are other related acids with the general formula...
4 KB (360 words) - 08:28, 27 August 2024
using sulfur trioxide. The reaction proceeds by initial formation of the pyrosulfate: 2 SO3 + ROH → ROSO2−O−SO3H ROSO2−O−SO3H → ROSO3H + SO3 Several million...
28 KB (3,064 words) - 17:12, 5 May 2024
Peroxomonosulfate SO2−4 Sulfate SO2−3 Sulfite S2O2−8 Peroxydisulfate S2O2−7 Pyrosulfate S2O2−6 Dithionate S2O2−5 Metabisulfite S2O2−4 Dithionite S2O2−3 Thiosulfate...
17 KB (4,498 words) - 17:55, 8 July 2024
Related compounds Sodium bisulfate Sodium sulfite Sodium persulfate Sodium pyrosulfate Supplementary data page Sodium sulfate (data page) Except where otherwise...
28 KB (2,850 words) - 06:42, 8 September 2024
In this case anhydrous salts, such as copper(II) sulfate or potassium pyrosulfate, can also be added to sequester the water by forming hydrates, shifting...
12 KB (1,405 words) - 13:17, 26 August 2024
7778–80–5 K2S2O5 potassium metabisulfite 16731–55–8 K2S2O7 potassium pyrosulfate 7790–62–7 K2S2O8 potassium persulfate 7727–21–1 K2Se potassium selenide...
139 KB (120 words) - 17:07, 15 July 2024
sulfite pyrosulfite SO2− 3 S 2O2− 5 sulfate pyrosulfate SO2− 4 S 2O2− 7 phosphite pyrophosphite PO3− 3 P 2O4− 5 phosphate pyrophosphate PO3− 4 P 2O4−...
11 KB (773 words) - 03:36, 25 September 2024
using sulfur trioxide. The reaction proceeds by initial formation of the pyrosulfate: 2 SO3 + RCH2OH → RCH2OSO2−O−SO3H RCH2OSO2−O−SO3H → RCH2OSO3H + SO3 Several...
11 KB (1,074 words) - 21:50, 5 May 2024
acid (pyrosulfuric acid) H2SO4·nSO3 +6 Disulfate (commonly known as pyrosulfate), S 2O2− 7 and trisulfate, S 3O2− 10 Pure disulfuric acid melts at 36 °C...
5 KB (137 words) - 14:25, 4 January 2024
angle at the C-O-C is larger. Tricarbonate Peroxodicarbonate Oxalate Pyrosulfate Peroxydisulfate Dithionate Trithionate Tetrathionate Pyrophosphate Polyphosphate...
6 KB (565 words) - 20:51, 30 June 2024
number of 4 and in addition to the tetrahedral sulfate ion, SO2− 4, the pyrosulfate, S 2O2− 7, trisulfate, S 3O2− 10 and pentasulfate S 5O2− 16 ions all...
11 KB (1,286 words) - 23:13, 8 July 2024
antimony oxides, while a higher concentration will produce antimony(III) pyrosulfate. The reaction of elemental antimony and 18 M sulfuric acid will also...
6 KB (402 words) - 22:13, 12 May 2024
elongated prisms (K+)4(PO2F−2)2(S2O2−7) Tetrapotassium difluorophosphate pyrosulfate C2/c: a = 13.00 Å, b = 7.543 Å, c = 19.01 Å, β = 130.07°, Z = 4 Ca(PO2F2)2·CH3COOCH2CH3...
45 KB (2,745 words) - 14:54, 9 July 2024
more side-products. Methanesulfonyl chloride Disulfuric acid Sodium pyrosulfate Acetic anhydride Trifluoromethanesulfonic anhydride Wachtmeister, C....
9 KB (728 words) - 12:50, 2 September 2024
00, b = 7.543, c = 19.01, β = 130.07°, Z = 4 1426.5 2.489 colourless; pyrosulfate + difluorophosphate K3[O3SOPO2OSO3] H1−xTi2(PO4)3−x(SO4)x (x=0.5–1) Na2MgTi(SO4)(PO4)2...
46 KB (2,907 words) - 13:48, 5 December 2023
"Physico-chemical properties and transition metal complex formation in alkali pyrosulfate and hydrogen sulfate melts". VII International Conference on Molten Slags...
4 KB (451 words) - 15:33, 28 May 2022