Enthalpy (/ˈɛnθəlpi/ ) is the sum of a thermodynamic system's internal energy and the product of its pressure and volume. It is a state function in thermodynamics...
48 KB (6,203 words) - 21:25, 4 July 2024
the enthalpy of vaporization (symbol ∆Hvap), also known as the (latent) heat of vaporization or heat of evaporation, is the amount of energy (enthalpy) that...
21 KB (1,015 words) - 20:23, 18 December 2023
thermodynamics, the standard enthalpy of formation or standard heat of formation of a compound is the change of enthalpy during the formation of 1 mole...
29 KB (1,880 words) - 10:38, 30 June 2024
In chemistry, the enthalpy of atomization (also atomisation in British English) is the enthalpy change that accompanies the total separation of all atoms...
2 KB (257 words) - 16:04, 11 July 2024
In thermodynamics, the enthalpy of mixing (also heat of mixing and excess enthalpy) is the enthalpy liberated or absorbed from a substance upon mixing...
12 KB (1,892 words) - 02:39, 17 May 2024
Bond-dissociation energy (redirect from Bond dissociation enthalpy)
the standard enthalpy change when A−B is cleaved by homolysis to give fragments A and B, which are usually radical species. The enthalpy change is temperature-dependent...
23 KB (2,316 words) - 12:13, 9 July 2024
In thermodynamics, the enthalpy of fusion of a substance, also known as (latent) heat of fusion, is the change in its enthalpy resulting from providing...
12 KB (1,702 words) - 18:24, 2 July 2024
The standard enthalpy of reaction (denoted Δ H reaction ⊖ {\displaystyle \Delta H_{\text{reaction}}^{\ominus }} ) for a chemical reaction is the difference...
16 KB (2,545 words) - 14:15, 2 October 2023
In thermodynamics, the enthalpy of sublimation, or heat of sublimation, is the heat required to sublimate (change from solid to gas) one mole of a substance...
3 KB (152 words) - 03:51, 10 March 2024
In thermochemistry, the enthalpy of solution (heat of solution or enthalpy of solvation) is the enthalpy change associated with the dissolution of a substance...
5 KB (681 words) - 12:45, 26 March 2024
Heat of combustion (redirect from Standard enthalpy change of combustion)
fuel energy/mass of fuel energy/volume of the fuel There are two kinds of enthalpy of combustion, called high(er) and low(er) heat(ing) value, depending on...
26 KB (2,645 words) - 12:43, 25 June 2024
stagnation enthalpy of a fluid is the static enthalpy of the fluid at a stagnation point. The stagnation enthalpy is also called total enthalpy. At a point...
4 KB (634 words) - 16:33, 29 May 2023
Hess's law (section Use of enthalpies of formation)
and physician who published it in 1840. The law states that the total enthalpy change during the complete course of a chemical reaction is independent...
11 KB (1,400 words) - 04:10, 25 June 2024
An enthalpy–entropy chart, also known as the H–S chart or Mollier diagram, plots the total heat against entropy, describing the enthalpy of a thermodynamic...
7 KB (804 words) - 05:15, 6 March 2024
Hydration energy (redirect from Hydration enthalpy)
In chemistry, hydration energy (also hydration enthalpy) is the amount of energy released when one mole of ions undergoes hydration. Hydration energy is...
3 KB (345 words) - 16:35, 28 September 2023
In chemistry and thermodynamics, the enthalpy of neutralization (ΔHn) is the change in enthalpy that occurs when one equivalent of an acid and a base undergo...
3 KB (370 words) - 10:39, 2 December 2022
performance. Since it is a constant-enthalpy process, it can be used to experimentally measure the lines of constant enthalpy (isenthalps) on the ( p , T )...
33 KB (4,460 words) - 13:20, 17 July 2024
Lattice energy (redirect from Lattice enthalpy)
Born–Haber cycle Chemical bond Madelung constant Ionic conductivity Enthalpy of melting Enthalpy change of solution Heat of dilution Atkins; et al. (2010). Shriver...
10 KB (1,205 words) - 20:26, 20 April 2024
Latent heat (redirect from Enthalpy of transformation)
as the liquid's sensible heat onto the surface. The large value of the enthalpy of condensation of water vapor is the reason that steam is a far more effective...
12 KB (1,547 words) - 12:58, 1 June 2024
an exothermic reaction is a "reaction for which the overall standard enthalpy change ΔH⚬ is negative." Exothermic reactions usually release heat. The...
5 KB (552 words) - 07:35, 19 March 2024
In thermodynamics, enthalpy–entropy compensation is a specific example of the compensation effect. The compensation effect refers to the behavior of a...
19 KB (2,790 words) - 14:44, 7 March 2024
Wind tunnel (redirect from High enthalpy wind tunnel)
transonic wind tunnel Supersonic wind tunnel Hypersonic wind tunnel High enthalpy wind tunnel Wind tunnels are also classified by the orientation of air...
49 KB (6,781 words) - 02:39, 19 July 2024
chemical reaction to the change in temperature, T, given the standard enthalpy change, ΔrH⊖, for the process. The subscript r {\displaystyle r} means...
22 KB (3,074 words) - 18:57, 8 July 2024
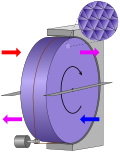
Thermal wheel (redirect from Enthalpy wheel)
thermal wheel, also known as a rotary heat exchanger, or rotary air-to-air enthalpy wheel, energy recovery wheel, or heat recovery wheel, is a type of energy...
15 KB (2,089 words) - 00:50, 20 May 2024
chemical bond. It is sometimes called the mean bond, bond enthalpy, average bond enthalpy, or bond strength. IUPAC defines bond energy as the average...
9 KB (1,318 words) - 02:44, 29 April 2024
Gibbs free energy (redirect from Free enthalpy)
{\textstyle U} is the internal energy of the system H {\textstyle H} is the enthalpy of the system S {\textstyle S} is the entropy of the system T {\textstyle...
33 KB (4,546 words) - 15:39, 17 July 2024
lattice energy (or more precisely enthalpy), which cannot otherwise be measured directly. The lattice enthalpy is the enthalpy change involved in the formation...
7 KB (941 words) - 22:25, 17 December 2023
Psychrometrics (section Specific enthalpy)
Mollier diagram coordinates are enthalpy and humidity ratio. The enthalpy coordinate is skewed and the lines of constant enthalpy are parallel and evenly spaced...
23 KB (3,228 words) - 01:34, 30 June 2024
change. The enthalpy of sublimation (also called heat of sublimation) can be calculated by adding the enthalpy of fusion and the enthalpy of vaporization...
23 KB (2,359 words) - 15:36, 20 June 2024
"The Vaporization Enthalpy of a Peculiar Pakistani Family" is a short science fiction story by Pakistani author Usman T. Malik. Inspired by Sufi poetry...
6 KB (717 words) - 15:29, 7 July 2024