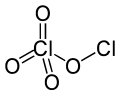
Chlorine perchlorate is a chemical compound with the formula Cl2O4. This chlorine oxide is an asymmetric oxide, with one chlorine atom in +1 oxidation...
6 KB (415 words) - 19:49, 19 March 2024
A perchlorate is a chemical compound containing the perchlorate ion, ClO−4, the conjugate base of perchloric acid (ionic perchlorate). As counterions,...
70 KB (7,786 words) - 13:48, 1 June 2024
chloryl perchlorate, Cl2O6 or [ClO2]+[ClO4]−, chlorine (V,VII) oxide dichlorine heptoxide, Cl2O7, chlorine (VII) oxide dichlorine octoxide, chlorine (VII)...
2 KB (286 words) - 00:10, 30 November 2023
Dichlorine hexoxide (redirect from Chloryl perchlorate)
However, in liquid or solid form, this chlorine oxide ionizes into the dark red ionic compound chloryl perchlorate [ClO 2]+ [ClO 4]− , which may be thought...
6 KB (514 words) - 14:14, 19 April 2024
(Cl2O6). Chlorine perchlorate may also be considered a chlorine derivative of perchloric acid (HOClO3), similar to the thermally unstable chlorine derivatives...
117 KB (13,009 words) - 22:07, 21 June 2024
Ammonium perchlorate MSDS. Sigma-Aldrich Helmut Vogt, Jan Balej, John E. Bennett, Peter Wintzer, Saeed Akbar Sheikh, Patrizio Gallone "Chlorine Oxides and...
8 KB (632 words) - 16:51, 12 June 2024
Sodium perchlorate is an inorganic compound with the chemical formula NaClO4. It consists of sodium cations Na+ and perchlorate anions ClO−4. It is a white...
9 KB (685 words) - 18:18, 23 June 2024
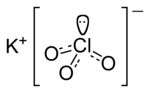
Potassium perchlorate is the inorganic salt with the chemical formula KClO4. Like other perchlorates, this salt is a strong oxidizer when the solid is...
17 KB (1,836 words) - 00:01, 22 May 2024
Perchloric acid (redirect from Hydrogen perchlorate)
salts, entails anodic oxidation of aqueous chlorine at a platinum electrode. Treatment of barium perchlorate with sulfuric acid precipitates barium sulfate...
13 KB (1,150 words) - 21:40, 7 June 2024
Lithium perchlorate is the inorganic compound with the formula LiClO4. This white or colourless crystalline salt is noteworthy for its high solubility...
10 KB (634 words) - 10:42, 17 June 2023
Sodium hypochlorite (redirect from Free Chlorine)
NaClO). It is commonly known in a dilute aqueous solution as bleach or chlorine bleach. It is the sodium salt of hypochlorous acid, consisting of sodium...
55 KB (5,761 words) - 07:27, 1 June 2024
Dichlorine heptoxide (redirect from Chlorine(VII) oxide)
Dichlorine heptoxide is the chemical compound with the formula Cl2O7. This chlorine oxide is the anhydride of perchloric acid. It is produced by the careful...
7 KB (592 words) - 03:31, 6 March 2024
a single bond to a perchlorate group, CH3−O−Cl(=O)3, in which chlorine has an oxidation state of +7. Like many other perchlorates, it is a high energy...
2 KB (111 words) - 18:22, 25 December 2023
pentafluoride – ClF5 Chlorine perchlorate – Cl2O4 Chlorine tetroxide – O3ClOOClO3 Chlorine trifluoride – ClF3 Chlorine trifluoride – ClF3 Chlorine trioxide – ClO3...
119 KB (8,726 words) - 04:34, 18 April 2024
the active ingredient in chlorine bleach), chlorine dioxide (ClO2), chlorate (ClO− 3), and perchlorate (ClO− 4). In terms of its acid–base properties...
18 KB (1,796 words) - 04:15, 9 May 2024
Fluorine perchlorate, also called perchloryl hypofluorite is the rarely encountered chemical compound of fluorine, chlorine, and oxygen with the chemical...
5 KB (506 words) - 21:29, 7 June 2024
Water purification (redirect from Chlorine dioxide disinfection)
remove impurities such as alkanes, perchlorates, and metals. Bioremediation has seen success because perchlorates are highly soluble, making them difficult...
67 KB (8,354 words) - 17:38, 8 June 2024
Nitrosyl perchlorate is the inorganic compound with the formula NO(ClO4). A hygroscopic white solid, it is the salt of the nitrosonium cation with the...
6 KB (512 words) - 20:35, 28 December 2023
Gomberg proposed a production method of chlorine tetroxide. He claimed that treating iodine and silver perchlorate in anhydrous diethyl ether produced it...
3 KB (308 words) - 23:07, 10 February 2024
Chlorate (category Chlorine oxides)
to perchlorate. It is suspected that chlorate and perchlorate may share a common natural formation mechanism and could be a part of the chlorine biogeochemistry...
8 KB (740 words) - 13:04, 24 April 2024
Chloralkali process (redirect from Alkali-chlorine process)
produce chlorine and sodium hydroxide (caustic soda), which are commodity chemicals required by industry. Thirty five million tons of chlorine were prepared...
16 KB (1,790 words) - 19:54, 16 January 2024
include. Ammonium perchlorate Ammonium nitrate Barium chlorate Potassium nitrate (saltpeter) Potassium chlorate Potassium perchlorate Strontium nitrate...
6 KB (472 words) - 03:26, 30 January 2024
Potassium chlorate is a compound containing potassium, chlorine and oxygen, with the molecular formula KClO3. In its pure form, it is a white crystalline...
15 KB (1,440 words) - 14:23, 11 June 2024
Shan Zhang (1985). "Chlorine trioxide, Cl2O6, a most efficient perchlorating reagent in new syntheses of anhydrous metal perchlorates, chloryl and nitryl...
6 KB (399 words) - 11:05, 12 May 2024
a hypothetical radical akin to chlorine dioxide The empirical formula for dichlorine hexoxide / chloryl perchlorate The perchloryl cation, ClO 3 + {\displaystyle...
331 bytes (74 words) - 09:38, 9 December 2021
67–68–5 ClF chlorine monofluoride 7790–89–8 ClF3 chlorine trifluoride 7790–91–2 ClF5 chlorine pentafluoride 13637–63–3 ClOClO3 chlorine perchlorate 27218–16–2...
139 KB (120 words) - 23:26, 30 April 2024
Hydronium perchlorate is an inorganic chemical compound with the chemical formula [H3O]ClO4. It is an unusual salt due to it being a solid and stable...
3 KB (203 words) - 20:29, 1 May 2024
Cheng Shan (1985). "Chlorine trioxide, Cl2O6, a most efficient perchlorating reagent in new syntheses of anhydrous metal perchlorates, chloryl and nitryl...
11 KB (959 words) - 16:49, 5 June 2024
drinking water by perchlorate. Perchlorate is also found in breast milk at significant levels, possibly attributable to perchlorate in drinking water...
62 KB (6,683 words) - 16:10, 23 June 2024
oxidation state of the central chlorine atom in this tetrahedral oxyanion is +7. In term of chemical kinetics, perchlorate is a non-labile species because...
7 KB (899 words) - 13:28, 23 May 2024