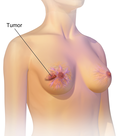
Palbociclib, sold under the brand name Ibrance among others, is a medication developed by Pfizer for the treatment of HR-positive and HER2-negative breast...
37 KB (3,558 words) - 20:55, 20 August 2024
additionally evaluate the efficacy and safety of inavolisib in combination with palbociclib and fulvestrant in patients that have PIK3CA-mutated, locally advanced...
21 KB (2,184 words) - 23:28, 5 August 2024
as has been shown in a study with rifampicin. Like the related drugs palbociclib and ribociclib, abemaciclib inhibits the enzymes cyclin-dependent kinase...
18 KB (1,467 words) - 23:44, 20 August 2024
cabozantinib, dasatinib, neratinib, eltrombopag, ibrutinib, lenvatinib, palbociclib, regorafenib, tofacitinib, and trelagliptin. BEACON Medicare Limited(BML)...
5 KB (232 words) - 10:23, 8 March 2024
HER2-negative advanced breast cancer in combination with abemaciclib or palbociclib in women with disease progression after endocrine therapy. It is given...
27 KB (2,341 words) - 12:46, 12 September 2024
overproliferation of cancer cells. The US FDA approved the first drug of this type, palbociclib (Ibrance), a CDK4/6 inhibitor, in February 2015, for use in postmenopausal...
24 KB (2,633 words) - 00:57, 22 April 2024
revenues), a pneumococcal conjugate vaccine ($6 billion in 2023 revenues), palbociclib ($4 billion in 2023 revenues), and tafamidis ($3 billion in 2023 revenues)...
168 KB (14,529 words) - 13:43, 16 September 2024
non-exclusive clinical supply agreement with Pfizer to provide palbociclib for its rintodestrant/palbociclib breast cancer trial. In July 2020, G1 entered into a...
20 KB (2,065 words) - 01:40, 29 December 2023
monotherapy and in combination with other drugs such as the CDK inhibitor palbociclib (Ibrance). Aromatase inhibitor Estrogen deprivation therapy Lee, Clara...
6 KB (530 words) - 16:44, 28 August 2024
therapeutic target for anti-tumor effectiveness. Three Cdk4/6 inhibitors – palbociclib, ribociclib, and abemaciclib – currently received FDA approval for clinical...
77 KB (9,017 words) - 19:41, 20 August 2024
therapies that are effective and have a manageable side effect profile. Palbociclib, one of the first CDK4/6 inhibitors approved by the FDA, has become essential...
35 KB (3,829 words) - 14:24, 17 June 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
61 KB (6,082 words) - 05:23, 17 June 2024
toremifene and fulvestrant, often used in combination with CDK4/6 inhibitors (palbociclib, ribociclib, or abemaciclib). When one endocrine therapy fails, most...
132 KB (14,919 words) - 03:41, 16 September 2024
teams has contributed to the development of two FDA-approved treatments; palbociclib (Breast Cancer Dream Team), and abraxane plus gemcitabine (Pancreatic...
15 KB (1,696 words) - 14:47, 20 November 2023
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
31 KB (2,908 words) - 03:40, 16 September 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
20 KB (1,809 words) - 14:07, 16 September 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
4 KB (411 words) - 21:05, 6 May 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
60 KB (6,439 words) - 04:38, 7 September 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
14 KB (1,105 words) - 06:03, 14 September 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
36 KB (3,299 words) - 19:46, 10 August 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
31 KB (2,821 words) - 10:12, 17 July 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
26 KB (2,640 words) - 19:37, 25 August 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
20 KB (1,915 words) - 00:12, 31 March 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
11 KB (729 words) - 05:53, 22 August 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
15 KB (1,438 words) - 02:57, 10 May 2024
pazopanib, vandetanib -ciclib Cyclin-dependent kinase 4/CDK6 inhibitors palbociclib, ribociclib -degib hedgehog signaling pathway inhibitors vismodegib,...
26 KB (2,150 words) - 12:06, 11 September 2024
and FDA clinical approval of three small molecule CDK4/6 inhibitors (Palbociclib (IBRANCE, Pfizer Inc. 2015), Ribociclib (KISQUALI, Novartis. 2017), and...
79 KB (9,502 words) - 07:42, 4 September 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
19 KB (1,681 words) - 07:11, 15 March 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
33 KB (2,959 words) - 02:09, 16 July 2024
Enzyme inhibitors FI (Tipifarnib§) CDK inhibitors (Abemaciclib Alvocidib† Palbociclib Ribociclib Seliciclib†) PrI Bortezomib Carfilzomib Oprozomib Ixazomib...
9 KB (595 words) - 19:48, 16 June 2024