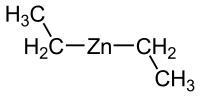Zinc iodide is the inorganic compound with the formula ZnI2. It exists both in anhydrous form and as a dihydrate. Both are white and readily absorb water...
7 KB (564 words) - 22:24, 6 March 2024
zinc chloride hydrates are heated, HCl gas evolves and hydroxychlorides result. In aqueous solution ZnCl2, as well as other halides (bromide, iodide)...
39 KB (3,996 words) - 21:33, 21 September 2024
Organozinc chemistry (section From zinc metal)
the first organozinc compound, diethylzinc, by heating ethyl iodide in the presence of zinc metal. This reaction produced a volatile colorless liquid that...
33 KB (3,394 words) - 06:05, 9 December 2023
are similar to those for zinc chloride, for which the toxic dose for humans is 3–5 g. Zinc chloride Zinc fluoride Zinc iodide Cadmium bromide F. Wagenknecht;...
7 KB (636 words) - 01:31, 20 July 2024
below 420 K with the zinc blende structure. Silver iodide is prepared by reaction of an iodide solution (e.g., potassium iodide) with a solution of silver...
8 KB (610 words) - 11:19, 23 September 2024
Iodomethylzinc iodide is the active reagent in the Simmons–Smith reaction. For example, iodomethylzinc iodide, formed in situ from diiodomethane and a zinc-copper...
865 bytes (74 words) - 17:50, 27 October 2019
Mercury(II) iodide is a chemical compound with the molecular formula HgI2. It is typically produced synthetically but can also be found in nature as the...
6 KB (414 words) - 20:28, 26 February 2024
Simmons–Smith reaction (section Non-zinc reagents)
diazomethane and zinc iodide. The reactivity of the system can also be increased by using the Furukawa modification, exchanging the zinc‑copper couple for...
27 KB (2,906 words) - 17:06, 26 September 2024
Copper(I) iodide is an inorganic compound with the chemical formula CuI. It is also known as cuprous iodide. It is useful in a variety of applications...
12 KB (1,128 words) - 10:02, 6 August 2024
ZnP2 Zinc fluoride – ZnF2 Zinc iodide – ZnI2 Zinc nitrate – Zn(NO3)2 Zinc oxide – ZnO Zinc phosphide – Zn3P2 Zinc pyrophosphate – Zn2P2O7 Zinc selenate...
119 KB (8,735 words) - 14:26, 16 September 2024
Diethylzinc (redirect from Diethyl zinc)
reaction of a 1:1 mixture of ethyl iodide and ethyl bromide with a zinc-copper couple, a source of reactive zinc. The compound crystallizes in a tetragonal...
12 KB (1,156 words) - 22:49, 26 August 2024
synthetic chemistry. It was first reported in 1848 from the reaction of zinc and ethyl iodide, and was the first compound known to contain a metal–carbon sigma...
143 KB (16,224 words) - 08:44, 27 August 2024
(0.8 equiv) of cadmium iodide (CdI2) is needed to promote the reaction. Alternatively, the use of cuprous bromide and zinc iodide sequentially as catalysts...
11 KB (1,298 words) - 17:16, 26 September 2024
Dimethylzinc (redirect from Dimethyl zinc)
methyl iodide on zinc or zinc-sodium alloy at elevated temperatures. 2 Zn + 2 CH3I → Zn(CH3)2 + ZnI2 Sodium assists the reaction of the zinc with the...
10 KB (864 words) - 22:58, 14 July 2024
Oligodynamic effect (section Zinc)
growth of algae. Copper- and zinc-treated shingles are available. Zinc iodide and zinc sulfate are used as topical antiseptics. Besides the individual toxic...
20 KB (2,135 words) - 09:19, 10 August 2024
interconnected by doubly bridging iodide ligands. One form consist of edge-sharing polytetrahedra. The other form resembles zinc iodide with interconnected adamantane-like...
7 KB (370 words) - 22:58, 10 August 2024
Radium iodide is an inorganic compound of radium and iodine with the chemical formula RaI2. It is the radium salt of hydrogen iodide, consisting of radium...
2 KB (97 words) - 21:19, 4 July 2024
Zinc–copper couple is an alloy of zinc and copper that is employed as a reagent in organic synthesis. The “couple” was popularized after the report by...
5 KB (684 words) - 21:49, 31 March 2024
form consist of edge-sharing polytetrahedra. The other form resembles zinc iodide with interconnected adamantane-like cages. In contrast, BeF2 is a 3-dimensional...
8 KB (570 words) - 22:15, 10 August 2024
Cadmium iodide is an inorganic compound with the formula CdI2. It is a white hygroscopic solid. It also can be obtained as a mono- and tetrahydrate. It...
6 KB (435 words) - 06:47, 2 July 2024
hydrocarbon mixture obtained by the condensation of methanol at 200 °C with a zinc iodide catalyst (the main component of the mixture being the isomer 2,2,3-trimethylbutane...
10 KB (913 words) - 22:20, 31 May 2023
7783–49–5 ZnH2 zinc hydride 14018–82–7 Zn(IO3)2 zinc iodate 7790–37–6 ZnI2 zinc iodide 10139–47–6 Zn(MnO4)2 zinc permanganate 23414–72–4 ZnMoO4 zinc molybdate...
139 KB (120 words) - 17:07, 15 July 2024
metals, notably samarium(II) iodide, chromium(II) chloride, titanium(II) chloride, cerium(III) halides such as cerium(III) iodide, and titanocene(III) chloride...
13 KB (1,269 words) - 19:55, 31 October 2023
Iodine (section Hydrogen iodide)
Ιώδης, meaning 'violet'. Iodine occurs in many oxidation states, including iodide (I−), iodate (IO− 3), and the various periodate anions. As the heaviest...
107 KB (11,980 words) - 14:46, 27 September 2024
dibromomethane or diazomethane and zinc iodide. The reactivity of the system can also be increased by exchanging the zinc‑copper couple for diethylzinc. Asymmetric...
14 KB (1,504 words) - 19:07, 5 July 2024
Bismuth(III) iodide is the inorganic compound with the formula BiI3. This gray-black salt is the product of the reaction of bismuth and iodine, which once...
7 KB (477 words) - 18:58, 12 May 2024
7783-49-5 Zn(IO3)2 zinc iodate 7790-37-6 ZnI2 zinc iodide 10139-47-6 ZnMoO4 zinc orthomolybdate Zn(NO2)2 zinc nitrite 10102-02-0 Zn(NO3)2 zinc nitrate 7779-88-6...
183 KB (107 words) - 22:11, 6 September 2024
Zinc iodate Zn(IO3)2·2H2O 0.07749 Zinc iodide ZnI2 430 432 445 467 490 510 Zinc nitrate Zn(NO3)2 98 138 211 Zinc oxalate ZnC2O4·2H2O 1.38×10−9 Zinc oxide...
84 KB (193 words) - 18:48, 2 September 2024
form consist of edge-sharing polytetrahedra. The other form resembles zinc iodide with interconnected adamantane-like cages. Beryllium compounds are toxic...
7 KB (404 words) - 22:18, 10 August 2024
halide (chloride, bromide, iodide), a carbonyl group and a metal. The reaction can be performed using magnesium, aluminium, zinc, indium, tin, samarium,...
6 KB (486 words) - 15:10, 4 January 2024