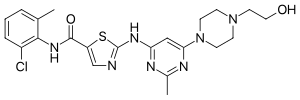
Imatinib, sold under the brand names Gleevec and Glivec (both marketed worldwide by Novartis) among others, is an oral targeted therapy medication used...
71 KB (6,702 words) - 05:32, 9 August 2024
Chronic myelogenous leukemia (section Imatinib)
action as a tyrosine kinase, targeted therapies (the first of which was imatinib) that specifically inhibit the activity of the BCR-ABL protein have been...
34 KB (3,877 words) - 01:25, 22 July 2024
patent imatinib mesylate in beta crystalline form (rather than imatinib or imatinib mesylate); thus they sought to prevent extant literature on imatinib or...
43 KB (5,292 words) - 19:42, 6 October 2024
kinase. Before the 2001 U.S. Food and Drug Administration (FDA) approval of imatinib, no drugs were available to alter the natural progression of CML. Only...
56 KB (6,475 words) - 07:21, 26 August 2024
the years. However, French scientists treating leukemia patients with imatinib, a drug used in treating cancer, noted an unexpected side effect: some...
16 KB (1,854 words) - 11:53, 6 October 2024
Tyrosine kinase (section GIST and Imatinib)
step for the development of cancer. Therefore, kinase inhibitors, such as imatinib and osimertinib, are often effective cancer treatments. Most tyrosine kinases...
43 KB (5,251 words) - 10:44, 24 June 2024
exon 17 are responsible for resistance to targeted therapy drugs like imatinib mesylate, a tyrosine kinase inhibitor. KIT-p.D419del (exon 8) — A subset...
43 KB (4,502 words) - 18:59, 16 July 2024
tissue. One of the most successful molecular targeted therapeutics is imatinib, marketed as Gleevec, which is a kinase inhibitor with exceptional affinity...
20 KB (2,234 words) - 01:21, 22 July 2024
anion, the spelling used is sometimes mesilate (as in imatinib mesilate, the mesylate salt of imatinib). Mesylate esters are a group of organic compounds...
4 KB (373 words) - 22:32, 16 February 2024
approved by the FDA for the treatment of renal cell carcinoma (RCC) and imatinib-resistant gastrointestinal stromal tumor (GIST) in January 2006. Sunitinib...
36 KB (3,447 words) - 05:37, 9 August 2024
small number of children also develop this disease. It is treated with imatinib (Gleevec in United States, Glivec in Europe) or other drugs. The five-year...
83 KB (8,161 words) - 00:29, 7 October 2024
CML CP) or Ph+ CML CP resistant or intolerant to prior therapy including imatinib. newly diagnosed Ph+ acute lymphoblastic leukaemia (ALL) in combination...
22 KB (1,994 words) - 04:41, 3 January 2024
GlaxoSmithKline in 2015 deal), carbamazepine (Tegretol), valsartan (Diovan), imatinib mesylate (Gleevec/Glivec), cyclosporine (Neoral/Sandimmune), letrozole...
144 KB (12,145 words) - 21:14, 28 September 2024
kinase inhibitors specific to such domains as CC, Y177, and Rho (such as imatinib and sunitinib) are important drugs against a variety of cancers including...
29 KB (3,538 words) - 15:01, 5 December 2023
anti-mast cell activity) Chronic myelogenous leukemia (largely replaced by imatinib, but still in use for its cost-effectiveness) Reported side effects are:...
20 KB (1,809 words) - 14:07, 16 September 2024
well as in accelerated and chronic phase CML that has not responded to imatinib. It is taken by mouth. Common side effects may include low platelets, low...
20 KB (1,697 words) - 00:44, 27 August 2024
return for a large sum of money. Due to patent protection, the Swiss drug imatinib is very expensive and cannot be afforded by most leukemia patients in China...
10 KB (798 words) - 11:06, 21 September 2024
reactions with phenelzine, and a potential interaction has been reported with imatinib, resulting in hepatotoxicity, and with lamotrigine. The common ginsengs...
32 KB (3,102 words) - 08:59, 2 October 2024
Topotecan Etoposide Teniposide Tafluposide Bortezomib Erlotinib Gefitinib Imatinib Vemurafenib Vismodegib Azacitidine Azathioprine Capecitabine Cladribine...
5 KB (324 words) - 12:07, 20 July 2024
cancer's cell division. The classic example of targeted development is imatinib mesylate (Gleevec), a small molecule which inhibits a signaling molecule...
31 KB (3,824 words) - 20:24, 15 August 2024
US FDA approved (imatinib mesylate) for the treatment of DFSP. As is true for all medicinal drugs with name ending in "ib," imatinib is a small molecular...
29 KB (3,131 words) - 12:39, 12 September 2024
eosinophils through ADCC antagonists of leukotriene synthesis or receptors imatinib (STI571) – inhibits PDGF-BB in hypereosinophilic leukemia Monoclonal antibodies...
29 KB (3,166 words) - 00:19, 9 September 2024
drug sensitivity), respond well or are anticipated to respond well to imatinib therapy as described for the treatment of diseases caused by FIP1L1-PDGFRA...
29 KB (3,354 words) - 17:01, 9 December 2023
rituximab, sirolimus, alefacept, and the tyrosine kinase inhibitors, imatinib, nilotinib, and dasatinib. Experimental therapies under investigation include...
38 KB (3,293 words) - 18:29, 29 August 2024
Charlotte F, Salvatierra J, Wechsler B, Graux C, et al. (June 2008). "Imatinib mesylate for platelet-derived growth factor receptor-beta-positive Erdheim-Chester...
22 KB (2,228 words) - 19:53, 23 August 2024
γ-secretase. Imatinib itself does not get into the brain so imatinib could not be used as an AD therapeutic. However it may be possible to identify imatinib-like...
8 KB (819 words) - 00:00, 4 March 2023
objective response rate with median progression free survival of 18 months Imatinib: 2% objective response rate with median progression free survival of 9...
21 KB (2,187 words) - 14:39, 28 May 2024
is now routine practice, as its presence indicates a likely response to imatinib, a tyrosine kinase inhibitor. Chusid et al. developed empirical diagnostic...
26 KB (2,491 words) - 20:07, 10 September 2024
inhibitor of Lyn and Bcr-Abl. It reached phase II clinical trials in 2010. Imatinib was the first Bcr-Abl tyrosine-kinase inhibitor and was highly successful...
8 KB (733 words) - 11:36, 12 August 2023
through which imatinib eliminates leukemic cells, that leukemic animals could be cured using imatinib, and that resistance to imatinib could be mediated...
6 KB (634 words) - 17:22, 22 August 2024