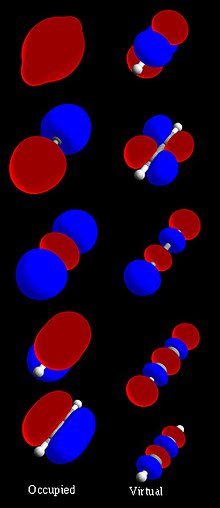
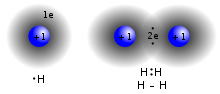
In quantum mechanics, an atomic orbital (/ˈɔːrbɪtəl/) is a function describing the location and wave-like behavior of an electron in an atom. This function...
84 KB (10,978 words) - 12:56, 20 October 2024
In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new hybrid orbitals (with different energies,...
33 KB (3,169 words) - 13:02, 22 September 2024
region. The terms atomic orbital and molecular orbital were introduced by Robert S. Mulliken in 1932 to mean one-electron orbital wave functions. At...
35 KB (4,390 words) - 11:31, 6 June 2024
which atomic orbital is combined in the term. The coefficients are the weights of the contributions of the n atomic orbitals to the molecular orbital. The...
6 KB (781 words) - 00:03, 6 April 2023
three main requirements for atomic orbital combinations to be suitable as approximate molecular orbitals. The atomic orbital combination must have the correct...
22 KB (2,959 words) - 13:06, 18 October 2024
orbital diagrams are diagrams of molecular orbital (MO) energy levels, shown as short horizontal lines in the center, flanked by constituent atomic orbital...
39 KB (4,968 words) - 12:08, 22 September 2024
Electron configuration (redirect from Orbital ordering)
molecule has a different orbital structure. The molecular orbitals are labelled according to their symmetry, rather than the atomic orbital labels used for atoms...
60 KB (6,147 words) - 04:46, 30 September 2024
Chemical bond (redirect from Atomic bond)
which includes orbital hybridization and resonance, and molecular orbital theory which includes the linear combination of atomic orbitals and ligand field...
40 KB (4,872 words) - 13:33, 22 September 2024
Sigma bond (redirect from Sigma molecular orbital)
overlapping of atomic orbitals. The concept of sigma bonding is extended to describe bonding interactions involving overlap of a single lobe of one orbital with...
8 KB (918 words) - 13:02, 22 September 2024
Azimuthal quantum number (redirect from Orbital quantum number)
quantum number for an atomic orbital that determines its orbital angular momentum and describes aspects of the angular shape of the orbital. The azimuthal quantum...
20 KB (2,143 words) - 19:17, 12 October 2024
Atom (redirect from Atomic chemical)
from the original on 7 December 2006. Manthey, David (2001). "Atomic Orbitals". Orbital Central. Archived from the original on 10 January 2008. Herter...
125 KB (12,855 words) - 14:56, 17 October 2024
original atomic level and one higher. The orbital which is in a lower energy state than the orbitals of the separate atoms is the bonding orbital, which...
7 KB (880 words) - 07:57, 10 May 2023
orbital (MO) theory, that were developed to use the methods of quantum mechanics to explain chemical bonding. It focuses on how the atomic orbitals of...
12 KB (1,519 words) - 13:06, 18 October 2024
molecular orbital theory a linear combination of atomic orbitals is performed first, followed by filling of the resulting molecular orbitals with electrons...
28 KB (3,673 words) - 05:58, 14 October 2024
molecular orbital). According to molecular orbital theory, molecular orbitals are often modeled by the linear combination of atomic orbitals. In a simple...
5 KB (660 words) - 20:31, 24 June 2023
which binds electrons in atoms. Some resemblance to atomic orbital models may be seen in a small atomic nucleus like that of helium-4, in which the two protons...
33 KB (4,076 words) - 22:52, 11 September 2024
Additionally, the cover art on three of their albums showcase stylised atomic orbitals. Orbital have been critically and commercially successful, known particularly...
36 KB (3,623 words) - 09:07, 7 October 2024
unoccupied s-orbital. Because an atom's s-orbital electrons are typically farthest from the nucleus, this results in a significant increase in atomic radius...
29 KB (1,865 words) - 04:36, 14 October 2024
An electron orbital may refer to: An atomic orbital, describing the behaviour of an electron in an atom A molecular orbital, describing the behaviour...
472 bytes (94 words) - 08:06, 27 October 2017
atomic orbital. The orbitals come in a variety of shapes—sphere, dumbbell, torus, etc.—with the nucleus in the middle. The shapes of atomic orbitals are...
70 KB (9,163 words) - 15:14, 12 October 2024
Core electron (redirect from Core orbital)
valence electrons can be described with atomic orbital theory. In atoms with a single electron the energy of an orbital is determined exclusively by the principal...
10 KB (1,396 words) - 17:09, 11 June 2024
basis atomic orbitals (AO) and molecular orbitals (MO): Atomic orbital → NAO → NHO → NBO → NLMO → Molecular orbital Natural (localized) orbitals are used...
5 KB (548 words) - 21:13, 16 October 2022
electron in a specific atomic orbital, which are functions representing 3 dimensional regions around the nucleus. The description of orbital motion as probability...
15 KB (2,641 words) - 01:18, 5 October 2024
basis function and atomic orbital are sometimes used interchangeably, although the basis functions are usually not true atomic orbitals. Within the basis...
36 KB (4,967 words) - 01:47, 12 October 2024
one-electron wave functions known as spin-orbitals. For an atomic orbital calculation, these are typically the orbitals for a hydrogen-like atom (an atom with...
31 KB (4,730 words) - 17:08, 7 October 2024
Slater-type orbitals (STOs) are functions used as atomic orbitals in the linear combination of atomic orbitals molecular orbital method. They are named...
15 KB (2,484 words) - 06:41, 17 November 2023
Look up orbital in Wiktionary, the free dictionary. Orbital may refer to: Atomic orbital Molecular orbital Hybrid orbital Orbit Earth orbit Orbit (anatomy)...
1 KB (178 words) - 11:34, 25 November 2023
Quantum number (redirect from Quantum numbers with spin-orbit interaction)
angular nodes present in an orbital. For example, for p orbitals, ℓ = 1 and thus the amount of angular nodes in a p orbital is 1. The magnetic quantum...
31 KB (3,125 words) - 15:04, 9 August 2024
Pi bond (redirect from Pi bonding molecular orbital)
orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbitals has...
7 KB (853 words) - 12:26, 22 September 2024
involved atomic orbital overlap six lobes of the other involved atomic orbital. This overlap leads to the formation of a bonding molecular orbital with three...
4 KB (356 words) - 07:00, 20 October 2024