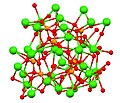
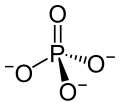
The term calcium phosphate refers to a family of materials and minerals containing calcium ions (Ca2+) together with inorganic phosphate anions. Some...
7 KB (602 words) - 03:25, 18 May 2024
Dicalcium phosphate is the calcium phosphate with the formula CaHPO4 and its dihydrate. The "di" prefix in the common name arises because the formation...
9 KB (721 words) - 00:55, 15 May 2024
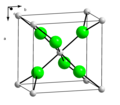
Tricalcium phosphate (sometimes abbreviated TCP), more commonly known as Calcium phosphate, is a calcium salt of phosphoric acid with the chemical formula...
18 KB (1,828 words) - 20:19, 24 April 2024
treating calcium hydroxide with phosphoric acid: Ca(OH)2 + 2 H3PO4 → Ca(H2PO4)2 + 2 H2O Samples of Ca(H2PO4)2 tend to convert to dicalcium phosphate: Ca(H2PO4)2...
7 KB (524 words) - 09:30, 11 June 2024
Amorphous calcium phosphate (ACP) is a glassy solid that is formed from the chemical decomposition of a mixture of dissolved phosphate and calcium salts (e...
12 KB (1,341 words) - 17:26, 1 December 2023
calcium carbonate, calcium citrate, calcium gluconate, calcium lactate, and calcium phosphate. The absorption of calcium from most food and commonly used...
37 KB (4,173 words) - 16:18, 26 April 2024
phosphate and form an insoluble compound. Calcium carbonate Calcium-based phosphate binders, such as calcium carbonate, directly decrease phosphate levels...
9 KB (655 words) - 08:06, 25 May 2024
occurrence of phosphates in biological systems is as the structural material of bone and teeth. These structures are made of crystalline calcium phosphate in the...
30 KB (2,574 words) - 09:12, 1 July 2024
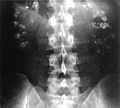
Kidney stone disease (redirect from Calcium stone)
stones are predominantly composed of calcium oxalate; struvite and calcium phosphate stones are less common. Calcium oxalate stones in children are associated...
132 KB (13,803 words) - 10:09, 14 June 2024
impure calcium sulfite, which oxidizes on storage to calcium sulfate. In the production of phosphoric acid from phosphate rock, calcium phosphate is treated...
19 KB (1,828 words) - 19:45, 7 June 2024
Parathyroid hormone (category Hormones of calcium metabolism)
enhances the uptake of phosphate from the intestine and bones into the blood. In the bone, slightly more calcium than phosphate is released from the breakdown...
34 KB (4,157 words) - 07:52, 21 May 2024
Milk (section Calcium phosphate structure)
notably calcium phosphate. It is present in excess and often, much greater excess of solubility of solid calcium phosphate. In addition to calcium, milk...
150 KB (15,507 words) - 20:58, 2 July 2024
Types of stones include struvite, calcium oxalate, urate, cystine, calcium phosphate, and silicate. Struvite and calcium oxalate stones are by far the most...
18 KB (2,216 words) - 11:47, 15 July 2024
Hyperparathyroidism (section Serum phosphate)
calcium reabsorption in the nephron. PTH also acts on the proximal convoluted tubule to decrease phosphate reabsorption to lower the serum phosphate....
53 KB (5,244 words) - 01:40, 17 May 2024
The original acid phosphate, made by Horsford's chemical company, and was a mixture of calcium, magnesium and potassium phosphate salts with a small...
5 KB (451 words) - 07:59, 16 July 2024
nephrocalcinosis is used to describe the deposition of both calcium oxalate and calcium phosphate. It may cause acute kidney injury. It is now more commonly used to...
15 KB (1,574 words) - 03:40, 17 September 2023
Hydroxyapatite (redirect from Calcium hydroxyapatite)
by phosphate anions in stoichiometric hydroxyapatite, are occupied by phosphate or hydrogen phosphate, HPO2−4, anions. Preparation of these calcium-deficient...
36 KB (3,775 words) - 20:18, 16 March 2024
contains in total approximately 1 kg, 99% in the skeleton in the form of calcium phosphate salts. The extracellular fluid (ECF) contains approximately 22 mmol...
30 KB (3,700 words) - 16:04, 11 May 2024
Calcific tendinitis (redirect from Calcium hydroxyapatite deposition disease)
Calcific tendinitis is a common condition where deposits of calcium phosphate form in a tendon, sometimes causing pain at the affected site. Deposits...
12 KB (1,364 words) - 15:21, 11 June 2024
Sarcoplasmic reticulum (section Calcium absorption)
changes (the calcium is said to be a second messenger). Calcium is used to make calcium carbonate (found in chalk) and calcium phosphate, two compounds...
13 KB (1,810 words) - 15:53, 11 May 2024
primarily of calcium phosphate crystals organized into four principal mineral phases, listed here in order of decreasing ratio of phosphate to calcium: whitlockite...
33 KB (3,758 words) - 17:17, 28 May 2024
Gene delivery (section Calcium phosphate)
pores or the damaged cell wall. Another simple methods involves using calcium phosphate to bind the DNA and then exposing it to cultured cells. The solution...
25 KB (2,877 words) - 17:15, 24 May 2024
with calcium chloride:[citation needed] CaCl2 + H4P2O7(aq) → Ca2P2O7·2 H2O + HCl. The anhydrous forms can be prepared by heating dicalcium phosphate:[citation...
8 KB (653 words) - 18:10, 29 June 2024
in a two-step process by treating dicalcium phosphate with sodium bisulfate, which precipitates calcium sulfate: CaHPO4 + NaHSO4 → NaH2PO4 + CaSO4 In...
7 KB (367 words) - 00:52, 15 May 2024
Phosphorus (section Phosphate rock)
important source of phosphates for fertiliser in the latter half of the 19th century. Phosphate rock, which usually contains calcium phosphate, was first used...
106 KB (12,394 words) - 03:27, 15 July 2024
of calcium lactate, calcium diphosphate, and tricalcium phosphate. The last is also used as a polishing agent in toothpaste and in antacids. Calcium lactobionate...
47 KB (5,900 words) - 02:56, 11 July 2024
levels of phosphate may rise (called hyperphosphatemia) leading to bone problems. Calcium acetate binds phosphate in the diet to lower blood phosphate levels...
8 KB (531 words) - 23:45, 3 March 2024
Microcalcifications in the breast are made up of calcium phosphate or calcium oxalate. When consisting of calcium phosphate, they are usually dystrophic calcifications...
4 KB (339 words) - 22:28, 8 December 2023
apples. Calcium carbonate is used therapeutically as phosphate binder in patients on maintenance haemodialysis. It is the most common form of phosphate binder...
79 KB (7,564 words) - 14:25, 12 July 2024
phosphate sources to give a solid precipitate of calcium phosphate: 3 CaCl2 + 2 PO3−4 → Ca3(PO4)2 + 6 Cl− Calcium chloride has a very high enthalpy change of...
36 KB (2,916 words) - 22:47, 9 July 2024