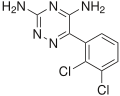
The FDA Adverse Event Reporting System (FAERS or AERS) is a computerized information database designed to support the U.S. Food and Drug Administration's...
5 KB (608 words) - 10:25, 22 July 2024
The Vaccine Adverse Event Reporting System (VAERS) is a United States program for vaccine safety, co-managed by the U.S. Centers for Disease Control and...
14 KB (1,485 words) - 22:56, 27 September 2023
FDA rolled out the FDA Adverse Event Reporting System (FAERS), replacing the earlier AERS system. FAERS is an online database that is used by the FDA...
27 KB (2,640 words) - 10:56, 22 August 2024
Administration (FDA) is responsible for monitoring post-marketing studies. The FDA has a reporting system called the FDA Adverse Event Reporting System, where...
32 KB (3,277 words) - 01:42, 12 August 2024
Pharmacovigilance (redirect from Adverse event reporting)
AE reporting: One of the fundamental principles of adverse event reporting is the determination of what constitutes an individual case safety report. During...
72 KB (9,242 words) - 06:50, 4 September 2024
disabling AE Grade 5 Death related to AE The FDA provides a database for reporting of adverse medical device events called the Manufacturer and User Facility...
9 KB (1,201 words) - 02:32, 12 September 2024
Pharmacovigilance FDA Adverse Event Reporting System (FAERS) Sakai T (2021). "医療ビッグデータにおける自発報告データベースの位置づけと利用可能性" [Role and Applicability of Spontaneous Reporting Databases...
994 bytes (91 words) - 22:01, 12 August 2023
Adverse Event Reporting Program.” It interacts with the FDA Adverse Event Reporting System (FAERS or AERS). MedWatch is used for reporting an adverse...
5 KB (513 words) - 08:03, 16 March 2023
as of 2013 only one case of adverse reaction to levosulpiride had been recorded on the FDA Adverse Event Reporting System Database. A case of rapid onset...
6 KB (316 words) - 08:08, 5 July 2024
SGLT2 inhibitor (section Adverse effects)
absolute risk is considered very low. In the FDA Adverse Event Reporting System an increase was reported in events of acute kidney injury associated with SGLT2...
44 KB (4,559 words) - 22:03, 7 July 2024
Cetirizine (section Adverse effects)
and Drug Administration (FDA) analyzed cases of pruritus after stopping cetirizine in the FDA Adverse Event Reporting System (FAERS) database and medical...
36 KB (3,347 words) - 11:03, 24 September 2024
sentinel events are recorded and published by the FDA's MedWatch program. Advantages of reporting sentinel events to The Joint Commission are: Adding to the...
6 KB (651 words) - 12:54, 23 September 2024
pancreatitis.According to the FDA’s Adverse Events Reporting System (FAERS), more than 150 patients taking Ozempic reported ileus or intestinal obstructions...
68 KB (5,684 words) - 04:12, 20 October 2024
Class of Neuropsychiatric Adverse Reactions to Mefloquine from Latent Class Modeling of FDA Adverse Event Reporting System Data". Drugs in R&D. 17 (1):...
2 KB (189 words) - 06:11, 7 July 2022
Dapagliflozin (section Adverse effects)
Inhibitors: A Pharmacovigilance Study with Data from the U.S. FDA Adverse Event Reporting System". Journal of Diabetes Research. 2020: 3695101. doi:10.1155/2020/3695101...
53 KB (4,975 words) - 03:43, 30 September 2024
Vestibulopathy (category Auditory system)
Class of Neuropsychiatric Adverse Reactions to Mefloquine from Latent Class Modeling of FDA Adverse Event Reporting System Data". Drugs in R&D. 17 (1):...
3 KB (338 words) - 07:25, 31 July 2024
Major adverse cardiovascular events (MACE, or major adverse cardiac events) is a composite endpoint frequently used in cardiovascular research. Despite...
13 KB (1,349 words) - 20:51, 4 February 2024
case reports, hundreds of additional cases of liver complications in people taking bicalutamide exist in the FDA Adverse Event Reporting System (FAERS)...
140 KB (15,744 words) - 18:51, 1 September 2024
sodium-channel blocking antiseizure medications. In March 2023, an FDA Adverse Event Reporting System analysis demonstrated signals of cardiac arrest, but not of...
65 KB (6,568 words) - 16:32, 18 October 2024
Vaccine Adverse Event Reporting System (VAERS) is a passive surveillance program administered jointly by the Food and Drug Administration (FDA) and the...
30 KB (3,078 words) - 20:12, 15 September 2024
Levofloxacin (section Adverse effects)
gravis exacerbation: evaluation of postmarketing reports from the US FDA adverse event reporting system and a literature review". Drug Safety. 34 (10):...
54 KB (5,402 words) - 21:33, 19 October 2024
Quinolone antibiotic (redirect from Adverse effects of fluoroquinolones)
treatment-related. Spontaneous reports to the U.S. FDA Adverse Effects Reporting System at the time of the 20 September 2011 U.S. FDA Pediatric Drugs Advisory...
65 KB (6,542 words) - 19:23, 19 October 2024
Artificial intelligence in healthcare (section US FDA)
electronic health records and/or adverse event reports. Organizations such as the FDA Adverse Event Reporting System (FAERS) and the World Health Organization's...
111 KB (12,985 words) - 06:19, 10 October 2024
Farmacovigilancia Española, Datos de Reacciones Adversas (redirect from Spanish pharmacovigilance system)
Pharmacovigilance System, is a pharmacovigilance database in Spain which was developed in 1982. Pharmacovigilance FDA Adverse Event Reporting System (FAERS) Montero...
1 KB (91 words) - 23:52, 19 July 2023
Quetiapine (section Adverse effects)
(2019). "Comparison of Quetiapine Abuse and Misuse Reports to the FDA Adverse Event Reporting System With Other Second-Generation Antipsychotics". Substance...
79 KB (6,427 words) - 19:30, 7 October 2024
Rivaroxaban (section Adverse effects)
highest number of reported cases of serious injury among regularly monitored medications to the FDA's Adverse Events Reporting System (AERS). In October...
29 KB (2,612 words) - 21:15, 28 September 2024
patients. In the United States several reporting systems have been built, such as the Vaccine Adverse Event Reporting System (VAERS), the Manufacturer and User...
36 KB (3,690 words) - 06:39, 1 September 2024
Indication (medicine) (section FDA approval)
now FDA-approved indication. Even after approval, the FDA CDER continues to do postmarking surveillance of the drug through MedWatch and FDA Adverse Event...
22 KB (2,346 words) - 04:28, 9 July 2024
Serious Risks/New Safety Information Identified by the FDA Adverse Event Reporting System (FAERS)". FDA. 3 January 2023. Archived from the original on 28 February...
85 KB (7,011 words) - 12:49, 10 October 2024
Serious Risks/New Safety Information Identified by the FDA Adverse Event Reporting System (FAERS)". FDA. Doraiswamy PM, Xiong GL (2006). "Pharmacological strategies...
41 KB (3,404 words) - 02:36, 27 August 2024