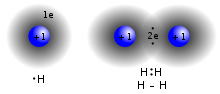
Lewis structures – also called Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDs) – are diagrams...
16 KB (2,140 words) - 19:28, 3 October 2024
be represented by a variety of diagrams called structural formulas. Lewis structures use a dot notation to represent the valence electrons for an atom;...
23 KB (2,209 words) - 12:13, 6 September 2024
Structural formula (redirect from Structure formula)
multiple types of ways to draw these structural formulas such as: Lewis structures, condensed formulas, skeletal formulas, Newman projections, Cyclohexane...
21 KB (2,596 words) - 11:08, 27 May 2024
Resonance (chemistry) (redirect from Resonance structure)
a chemical species can be described by a Lewis structure. For many chemical species, a single Lewis structure, consisting of atoms obeying the octet rule...
42 KB (5,094 words) - 06:04, 25 June 2024
Skeletal formula (redirect from Skeletal structure)
by the Lewis structure of molecules and their valence electrons. Hence they are sometimes termed Kekulé structures or Lewis–Kekulé structures. Skeletal...
28 KB (3,597 words) - 01:30, 6 July 2024
Natural bond orbital (section Lewis structures)
ideally close to 2.000, providing the most accurate possible “natural Lewis structure” of ψ. A high percentage of electron density (denoted %-ρL), often...
5 KB (548 words) - 21:13, 16 October 2022
Covalent bond (section Covalent structures)
unit of radiant energy). He introduced the Lewis notation or electron dot notation or Lewis dot structure, in which valence electrons (those in the outer...
28 KB (3,673 words) - 05:58, 14 October 2024
electrons between atoms, and was thus a model of ionic bonding. Both Lewis and Kossel structured their bonding models on that of Abegg's rule (1904). Although...
12 KB (1,519 words) - 13:06, 18 October 2024
that atom in a Lewis structure. When determining the best Lewis structure (or predominant resonance structure) for a molecule, the structure is chosen such...
9 KB (1,050 words) - 02:14, 19 September 2024
Triplet oxygen (section Lewis structure)
paramagnetic; i.e., it can be attracted to the poles of a magnet. Thus, the Lewis structure O=O with all electrons in pairs does not accurately represent the nature...
9 KB (894 words) - 07:03, 21 August 2024
several traditional and advanced bonding models such as simple Lewis and VSEPR structure, valence bond theory, molecular orbital theory, isovalent hybridization...
15 KB (1,822 words) - 11:34, 13 March 2023
Oxidation state (section Applied to a Lewis structure)
its Lewis structure. when the isolated tandem of a heteronuclear and a homonuclear bond leads to a bonding compromise in between two Lewis structures of...
46 KB (8,729 words) - 23:27, 12 October 2024
outermost electron shell of atoms. They can be identified by using a Lewis structure. Electron pairs are therefore considered lone pairs if two electrons...
23 KB (2,957 words) - 14:53, 17 October 2024
Octet rule (redirect from Lewis-Langmuir theory)
the Lewis structure with a double bond, but adds a question mark with the explanation that there is some doubt about the validity of this structure because...
22 KB (2,869 words) - 13:52, 4 October 2024
Lewis (1861), a sailing ship USS Lewis (DE-535), a destroyer escort in commission from 1944 to 1946 Lewis structure, a diagram of a molecule that shows...
4 KB (449 words) - 11:50, 11 October 2024
forms are merely alternative Lewis structure (valence bond theory) depictions of a single chemical species, whose true structure is a quantum superposition...
14 KB (1,474 words) - 04:25, 31 August 2024
either of the orbitals which overlap in the bonding process. As a Lewis structure, a single bond is denoted as AːA or A-A, for which A represents an...
5 KB (600 words) - 16:47, 9 October 2024
Molecular geometry (redirect from Molecular structure)
Commons has media related to Molecular geometry. Jemmis mno rules Lewis structure Molecular design software Molecular graphics Molecular mechanics Molecular...
23 KB (2,314 words) - 11:20, 14 September 2024
heuristic for rationalizing the structures of organic compounds. It gives a simple orbital picture equivalent to Lewis structures. Hybridisation theory is an...
33 KB (3,169 words) - 13:02, 22 September 2024
article by Lewis also introduced the concept of the electron pair in the covalent bond, the octet rule, and the now-called Lewis structure. Single covalent...
3 KB (412 words) - 19:05, 20 December 2022
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting...
22 KB (2,754 words) - 23:27, 18 October 2024
California, Berkeley. Lewis was best known for his discovery of the covalent bond and his concept of electron pairs; his Lewis dot structures and other contributions...
37 KB (4,207 words) - 13:51, 3 October 2024
(link) Glusker, Jenny Pickworth. (1994). Crystal structure analysis for chemists and biologists. Lewis, Mitchell., Rossi, Miriam. New York: VCH. ISBN 0-89573-273-4...
7 KB (849 words) - 02:55, 17 January 2024
Gene structure is the organisation of specialised sequence elements within a gene. Genes contain most of the information necessary for living cells to...
19 KB (2,320 words) - 17:06, 14 May 2024
equivalent conjugated contributing Lewis structures (the so-called Kekulé structures) that predominate. The true electronic structure is therefore a quantum-mechanical...
35 KB (4,303 words) - 03:15, 7 October 2024
conjugated system of bonds in its Lewis structure This disambiguation page lists articles associated with the title Lewis conjugate. If an internal link...
225 bytes (67 words) - 20:11, 28 December 2015
008. PMID 21111607. Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walters P (2002). "The Shape and Structure of Proteins". Molecular Biology of the Cell...
37 KB (4,204 words) - 19:21, 25 September 2024
Lewis' only his model assumed complete transfers of electrons between atoms, and was thus a model of ionic bonding. Both Lewis and Kossel structured their...
40 KB (4,872 words) - 13:33, 22 September 2024
the valence shell of a central atom is determined after drawing the Lewis structure of the molecule, and expanding it to show all bonding groups and lone...
45 KB (4,038 words) - 14:24, 26 September 2024