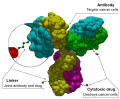
trials, a surrogate endpoint (or surrogate marker) is a measure of effect of a specific treatment that may correlate with a real clinical endpoint but does...
14 KB (1,681 words) - 17:15, 3 November 2024
powered. Surrogate endpoints are trial endpoints that have outcomes that substitute for a clinical endpoint, often because studying the clinical endpoint is...
15 KB (1,931 words) - 14:47, 16 July 2024
functions Gradient-enhanced kriging (GEK) OptiY Space mapping Surrogate endpoint Surrogate data Fitness approximation Computer experiment Conceptual model...
13 KB (1,519 words) - 20:53, 30 July 2024
measure that a drug is effective in treating the desired disease, and a surrogate endpoint is used to guide whether or not it is appropriate to proceed with...
16 KB (2,161 words) - 08:35, 16 June 2024
analysis of a surrogate endpoint at month twelve in a 54-month, randomized, double-blind placebo-controlled trial. The surrogate endpoint measured the...
8 KB (456 words) - 06:18, 6 October 2024
Cancer biomarker (section Surrogate endpoints)
before being brought to clinical trials. Some ideal characteristics of surrogate endpoint biomarkers include: Biomarker should be involved in process that causes...
46 KB (5,014 words) - 14:53, 4 August 2024
artist Allan McCollum Surrogate model, used in engineering design Surrogate endpoint, a measure of effect in clinical trials Surrogate key, a unique database...
3 KB (426 words) - 04:45, 10 June 2024
surrogate or intermediate endpoints. RMAT goes beyond breakthrough therapy features by allowing for accelerated approval of drugs based on surrogate endpoints...
28 KB (1,584 words) - 03:45, 11 August 2024
faster approval relies on use of surrogate endpoints. Drug approval typically requires clinical trials with endpoints that demonstrate a clinical benefit...
5 KB (550 words) - 17:52, 28 February 2024
outcome measures. Outcome measures can be divided into clinical endpoints and surrogate endpoints where the former is directly related to what the goal of the...
5 KB (525 words) - 17:32, 28 August 2024
supernatural Survivorship bias – Logical error, form of selection bias Surrogate endpoint – Biomarker proxy outcome measure Texas sharpshooter fallacy Woods...
6 KB (668 words) - 20:26, 8 November 2024
can are detected clinically. The biomarkers act as surrogate endpoints. The use of surrogate endpoints has been shown to significantly decrease the time...
13 KB (1,832 words) - 04:29, 28 October 2024
clinical endpoints are very costly as they have long durations and tend to need large numbers of patients. In contrast to clinical endpoints, surrogate endpoints...
61 KB (7,319 words) - 04:57, 25 November 2024
cardiovascular events. As such, the use of change in carotid IMT as a surrogate endpoint measure of drug efficacy in clinical trials, or in clinical management...
16 KB (1,952 words) - 16:58, 27 November 2023
intervals compared to baseline observations. CTCs are a potential surrogate endpoint in oncology trials, with reduced levels suggesting long-term clinical...
28 KB (3,411 words) - 03:40, 14 June 2024
States Food and Drug Administration approved DAAs on the basis of a surrogate endpoint called sustained virological response (SVR). SVR is achieved in a...
55 KB (6,733 words) - 21:23, 24 October 2024
gives separate definitions and prefers PFS. PFS is widely used as a surrogate endpoint in oncology. The definition of "progression" generally involves imaging...
7 KB (922 words) - 18:34, 16 May 2024
ozogamicin (trade name: Mylotarg) was approved based on a study with a surrogate endpoint, through the accelerated approval process. In June 2010, after evidence...
41 KB (4,305 words) - 11:56, 24 August 2024
standard chemotherapy. The accelerated approval was based on the surrogate endpoint of response rate. It was the first antibody-drug conjugate to be approved...
17 KB (1,318 words) - 04:34, 1 October 2024
reduction in the level of the biomarker alkaline phosphatase as a surrogate endpoint for clinical benefit. It is indicated for the treatment of primary...
19 KB (1,825 words) - 22:02, 12 July 2024
Treatment Interventions "Reporting of surrogate endpoints in randomised controlled trial reports (CONSORT-Surrogate): extension checklist with explanation...
87 KB (10,076 words) - 02:50, 13 November 2024
after 48 weeks of treatment with the drug or longer. The change was a surrogate endpoint and the trial did not establish clinical benefit of the drug, including...
13 KB (958 words) - 03:11, 6 July 2024
composite endpoints while performing relative effectiveness assessments. EUnetHTA: Endpoints used in Relative Effectiveness Assessment Surrogate Endpoints. This...
29 KB (3,027 words) - 20:22, 9 February 2024
2012. Fauber J, Chu E (27 October 2014). "The Slippery Slope: Is a Surrogate Endpoint Evidence of Efficacy?". MedPage Today. "Inlyta : EPAR - Product Information"...
16 KB (1,194 words) - 23:36, 16 August 2024
Biomarkers Definitions Working Group (March 2001). "Biomarkers and surrogate endpoints: preferred definitions and conceptual framework". Clinical Pharmacology...
38 KB (4,432 words) - 19:31, 11 November 2024
2 diabetes, as measured by glycated haemoglobin A1c (HbA1c) as a surrogate endpoint, similar to that of other oral antidiabetic drugs. The controversy...
57 KB (5,953 words) - 05:49, 12 October 2024
potential as an ALS biomarker (also known as a biological correlate or surrogate endpoint) by Prize4Life, a 501(c)(3) nonprofit organization dedicated to accelerating...
20 KB (2,523 words) - 10:25, 24 August 2023
(FDA) approves cancer therapies on the basis of arbitrarily assigned surrogate endpoints that typically have no bearing on overall survival or quality of...
3 KB (285 words) - 07:24, 19 November 2024
chemopreventive agents, in people and in animals. The relevance of ACF as a surrogate endpoint biomarker for cancer is controversial. Recently, several alternative...
3 KB (330 words) - 19:17, 31 December 2023
meet the primary endpoint. While imaging studies had shown that aducanumab did reduce amyloid plaque buildup, a surrogate endpoint for Alzheimer's treatment...
61 KB (4,888 words) - 02:50, 17 November 2024