A boronic acid is an organic compound related to boric acid (B(OH)3) in which one of the three hydroxyl groups (−OH) is replaced by an alkyl or aryl group...
27 KB (2,662 words) - 03:08, 9 November 2023
Protodeboronation (category Boron compounds)
metal-catalysed coupling reactions that utilise boronic acids (see Suzuki reaction). For a given boronic acid, the propensity to undergo protodeboronation...
11 KB (1,438 words) - 11:45, 14 February 2023
groups attached to boron. Phenylboronic acid is a white powder and is commonly used in organic synthesis. Boronic acids are mild Lewis acids which are generally...
9 KB (882 words) - 15:14, 22 May 2024
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula B(OH)3. It may also be called hydrogen orthoborate...
47 KB (4,999 words) - 08:56, 19 July 2024
other plants. Allotropes of boron Boron deficiency Boron oxide Boron nitride Boron neutron capture therapy Boronic acid Hydroboration-oxidation reaction...
119 KB (12,788 words) - 00:56, 10 July 2024
reactions include availability of common boronic acids, mild reaction conditions, and its less toxic nature. Boronic acids are less toxic and safer for the environment...
34 KB (3,851 words) - 03:55, 15 February 2024
Petasis reaction (category Chemical synthesis of amino acids)
multi-component reaction of an amine, a carbonyl, and a vinyl- or aryl-boronic acid to form substituted amines. Reported in 1993 by Nicos Petasis as a practical...
29 KB (3,232 words) - 12:16, 21 July 2024
Dynamic covalent chemistry (section Boronic acid)
natural systems. Boronic acid self-condensation or condensation with diols is a well-documented dynamic covalent reaction. The boronic acid condensation has...
18 KB (1,991 words) - 05:20, 1 December 2023
Organoboron chemistry (redirect from Carbon-boron bond)
BRn(OR)3-n are called borinic esters (n = 2), boronic esters (n = 1), and borates (n = 0). Boronic acids are key to the Suzuki reaction. Trimethyl borate...
26 KB (2,451 words) - 01:51, 23 January 2024
Functional group (section Groups containing boron)
Compounds containing boron exhibit unique chemistry due to their having partially filled octets and therefore acting as Lewis acids. note 1 Fluorine is...
31 KB (1,212 words) - 17:02, 13 July 2024
Chan–Evans–Lam coupling is a cross-coupling reaction between an aryl boronic acid and an alcohol or an amine to form the corresponding secondary aryl amines...
5 KB (621 words) - 13:31, 13 October 2023
organic reaction forming a new carbon–carbon bond from a thioester and a boronic acid using a metal catalyst. It is a cross-coupling reaction. This reaction...
12 KB (1,399 words) - 02:46, 6 March 2023
cores: Vaborbactam, used in combination with meropenem (Vabomere). Has a boronic acid core. Bacteria that can produce beta-lactamases include, but are not...
16 KB (1,696 words) - 16:20, 2 March 2024
Nevalainen T (November 2008). "Discovery of boronic acids as novel and potent inhibitors of fatty acid amide hydrolase". Journal of Medicinal Chemistry...
2 KB (154 words) - 11:39, 23 January 2023
Borylation (category Boron)
molecules. Boronic acids, and boronic esters are common boryl groups incorporated into organic molecules through borylation reactions. Boronic acids are trivalent...
35 KB (3,976 words) - 11:26, 22 April 2024
alternatives to boronic acids (RB(OH)2), boronate esters (RB(OR′)2), and organoboranes (R3B), particularly for Suzuki-Miyaura coupling. Boronic acids RB(OH)2...
5 KB (347 words) - 19:08, 1 June 2024
Carbonyl group (redirect from Carboxylic Acid Derivatives)
classes of organic compounds (such as aldehydes, ketones and carboxylic acids), as part of many larger functional groups. A compound containing a carbonyl...
9 KB (875 words) - 20:45, 15 July 2024
carboxylic acid is an organic acid that contains a carboxyl group (−C(=O)−OH) attached to an R-group. The general formula of a carboxylic acid is often...
23 KB (2,177 words) - 14:26, 21 July 2024
methods are not or only conditionally usable. In addition to the organic boronic acid derivatives, which often bind highly specifically to the 1,2-diol groups...
121 KB (12,792 words) - 07:51, 20 July 2024
peroxides are: Peroxy acids, the peroxy derivatives of many familiar acids, examples being peroxymonosulfuric acid and peracetic acid, and their salts, one...
5 KB (539 words) - 17:56, 16 July 2024
shown to conveniently detect peptides incorporating the boronic acid moiety by MALDI. Gentisic acid - Compound Summary, PubChem. Haynes, p. 5.91 Haynes,...
5 KB (415 words) - 18:01, 4 January 2024
Although ethers resist hydrolysis, they are cleaved by hydrobromic acid and hydroiodic acid. Hydrogen chloride cleaves ethers only slowly. Methyl ethers typically...
19 KB (1,835 words) - 03:40, 19 May 2024
protected aryl boronic ester which gives after acidic work-up the target product in 78% yield. The same reactants are forming with the aryl boronic ester at...
9 KB (908 words) - 12:26, 9 July 2023
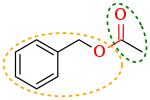
Ester (redirect from Carboxylic acid ester)
oxoacids (e.g. esters of acetic acid, carbonic acid, sulfuric acid, phosphoric acid, nitric acid, xanthic acid), but also from acids that do not contain oxygen...
42 KB (4,411 words) - 15:43, 18 July 2024
Ketone (section Acid/base properties of ketones)
Using very strong bases like lithium diisopropylamide (LDA, pKa of conjugate acid ~36) under non-equilibrating conditions (–78 °C, 1.1 equiv LDA in THF, ketone...
25 KB (2,948 words) - 23:08, 15 April 2024
Bortezomib (category Boronic acids)
which stands for pyrazinoic acid, phenylalanine and Leucine with a boronic acid instead of a carboxylic acid. The boron atom in bortezomib is proposed...
23 KB (2,216 words) - 22:24, 12 July 2024
In organic chemistry, sulfonic acid (or sulphonic acid) refers to a member of the class of organosulfur compounds with the general formula R−S(=O)2−OH...
15 KB (1,833 words) - 18:18, 15 July 2024
crystal As an acid catalyst in organic synthesis As a starting material for the production of other boron compounds, such as boron carbide Boron suboxide Boric...
15 KB (1,339 words) - 01:01, 5 March 2024
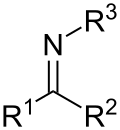
Imine (section Acid-base reactions)
carbon acids with nitroso compounds. The rearrangement of trityl N-haloamines in the Stieglitz rearrangement. By reaction of alkenes with hydrazoic acid in...
23 KB (2,633 words) - 07:34, 30 May 2024
forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds. The geometry of a molecule of BF3 is trigonal...
19 KB (1,848 words) - 02:21, 4 March 2024