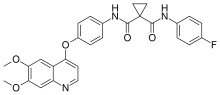
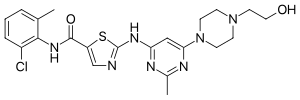
Cabozantinib, sold under the brand names Cometriq and Cabometyx among others, is an anti-cancer medication used to treat medullary thyroid cancer, renal...
21 KB (1,640 words) - 06:51, 18 September 2024
partnered its lead cancer drug candidate, XL-184 (which would become called cabozantinib) and another cancer candidate, XL-281, with Bristol Myers Squibb; BMS...
12 KB (1,034 words) - 18:16, 11 July 2024
surgery. Cabozantinib, trade name Cometriq, was granted marketing approval (November 2012) by the U.S. FDA for this indication. Cabozantinib which is...
19 KB (2,285 words) - 11:17, 14 October 2024
daclatasvir, sofosbuvir, afatinib, axitinib, brigatinib, baricitinib, cabozantinib, dasatinib, neratinib, eltrombopag, ibrutinib, lenvatinib, palbociclib...
5 KB (232 words) - 10:23, 8 March 2024
spasticity in children aged two years and older. In 2016, Ipsen licensed cabozantinib from Exelixis, which received marketing authorization the same year for...
23 KB (2,333 words) - 11:09, 11 August 2024
common adverse reaction to cytotoxic chemotherapy drugs, particularly cabozantinib, cytarabine, doxorubicin, and fluorouracil and its prodrug capecitabine...
16 KB (1,639 words) - 14:02, 28 August 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
121 KB (13,799 words) - 17:37, 8 October 2024
Institute. His work has led to the establishment of several novel drugs (cabozantinib, avelumab, pazopanib and others including combinations) and prognostic...
17 KB (1,497 words) - 16:35, 25 November 2023
cell carcinoma over everolimus in 2015 and was approved by the FDA. Cabozantinib also demonstrated an overall survival benefit over everolimus and was...
56 KB (5,187 words) - 04:02, 20 September 2024
that it is due to the decreased absorption of abacavir by orlistat. Cabozantinib: Drugs from the MRP2 inhibitor (Multidrug resistance-associated protein...
33 KB (2,771 words) - 17:59, 28 July 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
31 KB (3,430 words) - 05:24, 28 August 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
92 KB (8,729 words) - 10:10, 12 October 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
13 KB (1,177 words) - 02:19, 4 September 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
68 KB (5,999 words) - 20:41, 31 August 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
149 KB (15,702 words) - 00:28, 14 October 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
24 KB (1,710 words) - 19:18, 30 August 2024
c-Met inhibitors are currently[when?] in clinical trials. Crizotinib and cabozantinib were the first to be approved by the U.S. FDA. Crizotinib received accelerated...
32 KB (3,542 words) - 19:34, 2 July 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
10 KB (579 words) - 06:46, 26 June 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
25 KB (2,086 words) - 02:24, 16 July 2024
cancer (MTC) who require systemic therapy following prior treatment with cabozantinib and/or vandetanib. In September 2024, the FDA granted traditional approval...
26 KB (2,239 words) - 05:10, 15 October 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
18 KB (1,573 words) - 05:19, 15 October 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
42 KB (3,935 words) - 04:01, 30 April 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
110 KB (10,692 words) - 03:53, 30 September 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
26 KB (2,420 words) - 02:40, 26 September 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
34 KB (3,021 words) - 12:39, 5 August 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
22 KB (1,994 words) - 04:41, 3 January 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
69 KB (6,048 words) - 05:15, 1 October 2024
Clinical trial number NCT03141177 for "A Study of Nivolumab Combined With Cabozantinib Compared to Sunitinib in Previously Untreated Advanced or Metastatic...
46 KB (4,813 words) - 05:06, 15 October 2024
those who had tumor progression while on sorafenib compared to placebo. Cabozantinib, which is an inhibitor of multiple tyrosine kinases including VEGFR,...
90 KB (10,117 words) - 08:44, 9 October 2024
(PNB-0408) Kinase inhibitors: Altiratinib AM7 AMG-458 Amuvatinib BMS-777607 Cabozantinib Capmatinib Crizotinib Foretinib Golvatinib INCB28060 JNJ-38877605 K252a...
31 KB (2,651 words) - 05:34, 9 August 2024