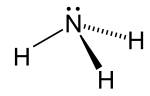
Stannane /ˈstæneɪn/ or tin hydride is an inorganic compound with the chemical formula SnH4. It is a colourless gas and the tin analogue of methane. Stannane...
3 KB (100 words) - 09:18, 12 April 2024
Organotin chemistry (redirect from Stannanes)
scientific study of the synthesis and properties of organotin compounds or stannanes, which are organometallic compounds containing tin–carbon bonds. The first...
24 KB (2,623 words) - 18:21, 19 September 2024
Stille reaction (section Stannane)
is widespread. In regards to limitations, both very bulky stannane reagents and stannanes with substitution on the α-carbon tend to react sluggishly...
47 KB (5,491 words) - 13:23, 22 February 2024
Fentin acetate (redirect from Stannane, acetoxytriphenyl-)
Fentin acetate Names IUPAC name (acetoxy)(triphenyl)stannane Other names Phentin acetate; Triphenyltin acetate; Triphenylstannyl acetate; Acetic acid tri(phenyl)stannyl...
4 KB (170 words) - 14:41, 16 September 2024
Organostannane addition (redirect from Allylic stannane addition)
number of acyclic transition states. (E)-Stannanes exhibit higher syn selectivity than the corresponding (Z)-stannanes. (3) In the presence of certain Lewis...
8 KB (971 words) - 16:58, 24 June 2022
Related alkanes Ethane Propane Butane Related compounds Silane Germane Stannane Plumbane Supplementary data page Methane (data page) Except where otherwise...
90 KB (8,771 words) - 16:01, 30 September 2024
alkynes, and all hydrocarbons silicon: silane germanium: germane tin: stannane lead: plumbane nitrogen: ammonia ("azane" when substituted), hydrazine...
21 KB (2,283 words) - 16:04, 30 September 2024
fluoroacetate Sodium pentachlorophenate Sodium selenate Sodium selenite Stannane, acetoxytriphenyl- Strychnine Strychnine sulfate Sulfotep Sulfoxide, 3-chloropropyl...
10 KB (825 words) - 19:18, 7 August 2024
germane GeH4 and digermane Ge2H6. They are germanium analogues of alkanes. Stannane SnH4, a strong reducing agent slowly decomposes at room temperature to...
9 KB (818 words) - 15:44, 25 June 2023
+4 oxidation states: tin(II) sulfide and tin(IV) sulfide (mosaic gold). Stannane (SnH 4), with tin in the +4 oxidation state, is unstable. Organotin hydrides...
80 KB (8,948 words) - 10:26, 23 September 2024
replaced by an aryl boronic acid and in the Stille reaction by an aryl stannane. The cycle also extends to the other group 10 element nickel for example...
13 KB (1,355 words) - 19:29, 15 December 2023
alkynes. The typical stannane is tributyltin hydride. The reaction mechanism is assumed to operate via oxidative addition of the stannane to give a stannyl...
2 KB (170 words) - 18:45, 13 November 2023
is unstable as compared to its lighter congeners silane, germane, and stannane. It cannot be made by methods used to synthesize GeH4 or SnH4. In 1999...
8 KB (723 words) - 09:44, 9 April 2024
−136 52 75-10-5 Difluorocarbamyl fluoride F2NCOF −52 −152.2 99 2368-32-3 Stannane SnH4 −51.8 −146 123 2406-52-2 Tetrafluoropropyne CF3C≡CF −50.39 112 20174-11-2...
95 KB (4,549 words) - 03:40, 24 September 2024
stable primary alkoxy stannane in solution. The reaction is then quenched irreversibly, with the less hindered primary alkoxy stannane reacting more rapidly...
29 KB (3,757 words) - 15:11, 13 February 2024
Retrieved 2021-04-21. TETRAVINYLTIN - ChemicalBook Tetravinylstannane at Encyclopedia of Reagents for Organic Synthesis Stannane, tetraethenyl at NIST WebBook...
4 KB (229 words) - 03:13, 22 January 2024
This biosynthesis can be mimicked in the laboratory by preparing allylic stannane from farnesol, termed Corey synthesis. There are diverse ways to synthesize...
16 KB (1,706 words) - 21:56, 3 September 2023
0564 Related compounds Related tetrahydride compounds Methane Germane Stannane Plumbane Related compounds Phenylsilane Vinylsilane Disilane Trisilane...
20 KB (2,208 words) - 16:40, 22 September 2024