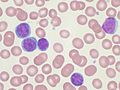
Ibrutinib, sold under the brand name Imbruvica among others, is a small molecule drug that inhibits B-cell proliferation and survival by irreversibly...
29 KB (2,605 words) - 06:34, 1 July 2024
acquire oncology firm Pharmacyclics and its treatment for blood cancers, ibrutinib; AstraZeneca had also been bidding to acquire Pharmacyclics. Under the...
39 KB (3,327 words) - 18:44, 14 July 2024
adverse effects relative to ibrutinib. In pre-clinical studies, it was shown to be more potent and selective than ibrutinib, the first-in-class BTK inhibitor...
14 KB (1,043 words) - 04:50, 1 July 2024
underlying disease. Ibrutinib is another agent that has been approved for use in this condition. Combination treatment with ibrutinib and rituximab showed...
59 KB (5,780 words) - 03:48, 13 July 2024
inhibits BTK in a way that is different from the prototypical BTK inhibitor ibrutinib by binding in a different way that avoids a genetic change (mutation at...
17 KB (1,163 words) - 14:36, 6 July 2024
well as potential for combination is expected from other drugs such as ibrutinib and idelalisib, both of which were also approved in 2014 to treat CLL...
27 KB (2,563 words) - 21:58, 1 July 2024
brigatinib, baricitinib, cabozantinib, dasatinib, neratinib, eltrombopag, ibrutinib, lenvatinib, palbociclib, regorafenib, tofacitinib, and trelagliptin....
5 KB (232 words) - 10:23, 8 March 2024
first line treatments may be offered. As of 2021, BTK inhibitors such as ibrutinib and acalabrutinib are often recommended for first line treatment of CLL...
72 KB (7,589 words) - 01:51, 25 June 2024
September 2021. Retrieved 2021-10-01. "Summary of opinion: Imbruvica,ibrutinib". Case Medical Research. 2019-06-28. doi:10.31525/cmr-1658a62. ISSN 2643-4652...
25 KB (1,860 words) - 23:03, 7 June 2024
pentostatin, etanercept, and alemtuzumab. In August 2017, the US FDA approved ibrutinib to treat chronic GvHD after failure of one or more other systemic treatments...
37 KB (4,234 words) - 22:42, 24 May 2024
randomized, active control, open-label trial, comparing zanubrutinib and ibrutinib in participants with MYD88 L265P mutation (MYD88MUT) WM. Participants...
23 KB (1,765 words) - 02:36, 28 April 2024
In January 2019, the US Food and Drug Administration (FDA) approved ibrutinib in combination with obinutuzumab for people with chronic lymphocytic leukemia/small...
17 KB (1,509 words) - 06:04, 5 July 2024
by CHOP, produced responses in 75% cases. A phase 1 clinical trial of ibrutinib – an inhibitor of Bruton's tyrosine kinase – in 13 patients reported responses...
15 KB (1,549 words) - 00:11, 19 May 2024
mechanistic step by many covalent inhibitor drugs. Cancer drugs such as ibrutinib, osimertinib, and rociletinib have an acrylamide functional group as a...
27 KB (2,825 words) - 08:27, 20 March 2024
cerebrospinal fluid. The 2-year EFS rate with ibrutinib was 80% (95% confidence interval [CI], 58%-91%), the 2-year ibrutinib survival rate was 81% (95% CI, 49%-94%)...
18 KB (2,181 words) - 20:15, 27 June 2024
CXCR4 WHIM mutations has been associated with clinical resistance to ibrutinib in patients with Waldenström's macroglobulinemia. While CXCR4's expression...
22 KB (2,660 words) - 15:08, 13 July 2024
chairman and CEO of Pharmacyclics. The company then focused its research on ibrutinib (branded as Imbruvica), a B cell cancer drug highly effective for treating...
25 KB (2,371 words) - 10:14, 11 July 2024
Confirms Sustained Superior Progression-free Survival of Zanubrutinib Versus Ibrutinib for Treatment of Relapsed/Refractory Chronic Lymphocytic Leukemia and...
25 KB (2,299 words) - 17:57, 21 May 2024
Guillain-Barre syndrome or the paralysis is a side effect of the clinical drug, Ibrutinib, he is taking to control his blood cancer. CIDP is a neurological disorder...
36 KB (5,021 words) - 23:56, 5 July 2024
would acquire Pharmacyclics, as well as its lead anti-cancer compound ibrutinib (Imbruvica) for $21 billion. As part of the deal, AbbVie will pay $261...
6 KB (321 words) - 18:21, 18 December 2023
Steven; Hurria, Arti; Brown, Jennifer R.; Lozanski, Gerard (2018-12-27). "Ibrutinib Regimens versus Chemoimmunotherapy in Older Patients with Untreated CLL"...
8 KB (800 words) - 15:49, 2 October 2023
Cobimetinib Selumetinib Trametinib EML4-ALK Crizotinib Entrectinib Lorlatinib Bruton's Acalabrutinib Ibrutinib Orelabrutinib Pirtobrutinib Zanubrutinib...
12 KB (760 words) - 05:52, 10 June 2024
Two Bruton tyrosine kinase inhibitors (BTKi), one In November 2013, ibrutinib (brand name Imbruvica, Pharmacyclics LLC) and one in October 2017, acalabrutinib...
41 KB (4,549 words) - 08:59, 25 January 2024
considered to be disease-causing mutations. Approved drugs that inhibit BTK: Ibrutinib (Imbruvica), a selective Bruton's tyrosine kinase inhibitor. Acalabrutinib...
22 KB (2,389 words) - 06:43, 22 April 2024
Axitinib L01EK02 Cediranib L01EK03 Tivozanib L01EK04 Fruquintinib L01EL01 Ibrutinib L01EL02 Acalabrutinib L01EL03 Zanubrutinib L01EL04 Orelabrutinib L01EL05...
12 KB (877 words) - 15:36, 25 January 2024
Pharmaceuticals metastatic non-small cell lung cancer with BRAF V600E mutation Ibrutinib Pharmacyclics chronic graft-versus-host disease Glecaprevir/pibrentasvir...
34 KB (180 words) - 22:09, 8 April 2024
Woyach, Jennifer A. (1 October 2018). "The BTK Inhibitor ARQ 531 Targets Ibrutinib-Resistant CLL and Richter Transformation". Cancer Discovery. 8 (10): 1300–1315...
7 KB (501 words) - 19:50, 24 April 2024
infused back into the donor patient); 3) Bruon's tyrosine kinase inhibitor, ibrutinib, to block the B-cell maturating actions of this kianase; 4) BCL inhibitor...
67 KB (7,967 words) - 19:22, 29 April 2024
Richter transformation: A mimic of acute myeloid leukemia that responded to Ibrutinib monotherapy". American Journal of Hematology. 95 (10): 1221–1223. doi:10...
37 KB (4,783 words) - 14:57, 23 February 2024
January 19, 2017, the FDA approved the first ever targeted drug for MZL, ibrutinib. This drug works by inhibiting Bruton's tyrosine kinase (BKT), which is...
116 KB (14,599 words) - 20:09, 24 January 2024