Cyanostar
A cyanostar (pentacyanopentabenzo[25]annulene) is a shape-persistent macrocycle that binds anions.[1][2]
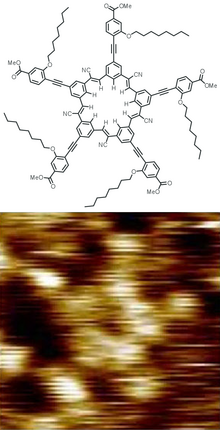
Synthesis
[edit]The cyanostar structure is synthesized in a one-pot process among five equivalents of a benzaldehyde bearing a meta-cyanomethyl substituent. A series of Knoevenagel condensation reactions catalyzed by various bases stitches them together to make the C5-symmetric structure.[3]
Anion binding
[edit]Cyanostar binds anions through hydrogen bonding from the C–H bonds, as the hydrogen has a positive electrostatic potential.[3] It is the first binder to make use of cyanostilbene's electropositive CH groups. The CH bonds create an electropositive region in the center of the macrocycle, creating a binding pocket. Cyanostar strongly binds anions that usually can only be bound weakly. The increased binding arises from the formation of a 2:1 complex, with two cyanostars sandwiching the anion on each side.[3] An extended version of this structural pattern is a 4:3 alternating stack of cyanostar molecules complexing a hydrogen-bonded chain of dihydrogen phosphate units.[4]
Rotaxanes
[edit]Two cyanostars can be threaded onto a phosphate diester structure, forming a rotaxane. Because they have a high affinity for the central phosphate group only when it is in its anionic form, there is a substantial and reversible structural change in response to acid–base changes in solution.[5]
References
[edit]- ^ Liu, Yun; Singharoy, Abhishek; Mayne, Christopher G.; Sengupta, Arkajyoti; Raghavachari, Krishnan; Schulten, Klaus; Flood, Amar H. (2016). "Flexibility Coexists with Shape-Persistence in Cyanostar Macrocycles". J. Am. Chem. Soc. 138 (14): 4843–4851. doi:10.1021/jacs.6b00712. PMC 4957974. PMID 27014837.
- ^ Zahran, EM; Fatila, EM; Chen, CH; Flood, AH; Bachas, LG (2018). "Cyanostar: C-H Hydrogen Bonding Neutral Carrier Scaffold for Anion-Selective Sensors". Anal. Chem. 90 (3): 1925–1933. doi:10.1021/acs.analchem.7b04008. PMID 29356501.
- ^ a b c Lee, Semin; Chen, Chun-Hsing; Flood, Amar H. (2013). "A Pentagonal Cyanostar Macrocycle with Cyanostilbene CH Donors Binds Anions and Forms Dialkylphosphate [3]Rotaxanes". Nature Chemistry. 5 (8): 704–710. Bibcode:2013NatCh...5..704L. doi:10.1038/nchem.1668. PMID 23881503.
- ^ Fatila, Elisabeth M.; Pink, Maren; Twum, Eric B.; Kartya, Jonathan A.; Flood, Amar H. (2018). "Phosphate–phosphate oligomerization drives higher order co-assemblies with stacks of cyanostar macrocycles". Chem. Sci. 9 (11): 2863–2872. doi:10.1039/C7SC05290A. PMC 5941797. PMID 29780454.
- ^ Qiao, Bo; Liu, Yun; Lee, Semin; Pink, Maren; Flood, Amar H. (2016). "High-yield Synthesis and Acid–Base Response of Phosphate-Templated [3]Rotaxanes". Chem. Commun. 52 (94): 13675–13678. doi:10.1039/C6CC08113D. PMID 27812564. S2CID 37747341.


 French
French Deutsch
Deutsch