Organochromium chemistry

Organochromium chemistry is a branch of organometallic chemistry that deals with organic compounds containing a chromium to carbon bond and their reactions.[1][2] The field is of some relevance to organic synthesis. The relevant oxidation states for organochromium complexes encompass the entire range of possible oxidation states from –4 (d10) in Na4[Cr–IV(CO)4] to +6 (d0) in oxo-alkyl complexes like Cp*CrVI(=O)2Me.
History
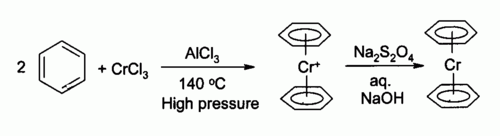
[edit]The first organochromium compound was described in 1919 by Franz Hein.[3] He treated phenylmagnesium bromide with chromium(III) chloride to give a new product (after hydrolysis) which he incorrectly identified as pentaphenyl chromium bromide (Ph5CrBr). Years later, in 1957 H.H. Zeiss et al. repeated Hein's experiments and correctly arrived at a cationic bisarene chromium sandwich compound (ArH2Cr+).[4] Bis(benzene)chromium itself was discovered around the same time in 1956 by Ernst Otto Fischer by reaction of chromium(III) chloride, benzene and aluminum chloride.[5][6] The related compound chromocene had been discovered a few years earlier in 1953 also by Fischer.[7]
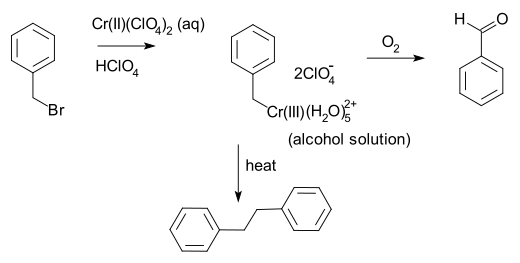
Anet and Leblanc also in 1957 prepared a benzyl chromium solution from benzyl bromide and chromium(II) perchlorate.[8] This reaction involves one-electron oxidative addition of the carbon-bromine bond, a process which was shown by Kochi[9][10] to be a case of double single electron transfer, first to give the benzyl free radical and then to the benzyl anion.
G. Wilke et al. introduced tris-(η-allyl)chromium in 1963 as an early Ziegler–Natta catalyst, albeit of limited commercial success.[11][12] Chromocene compounds were first employed in ethylene polymerization in 1972 by Union Carbide[13] and continue to be used today in the industrial production of high-density polyethylene.
The organochromium compound (phenylmethoxycarbene)pentacarbonylchromium, Ph(OCH3)C=Cr(CO)5 was the first carbene complex to be crystallographically characterized by Fischer in 1967 (now called a Fischer carbene).[14] The first ever carbyne, this one also containing chromium, made its debut in 1973.[15]
The first example of a proposed metal-metal quintuple bond is found in a compound of the type [CrAr]2, where Ar is a bulky aryl ligand.
Applications in organic synthesis
[edit]Although organochromium chemistry is heavily employed in industrial catalysis, relatively few reagents have been developed for applications in organic synthesis. Two are the Nozaki-Hiyama-Kishi reaction (1977) (transmetallation with organonickel intermediate) and the Takai olefination (1986)(oxidation of Cr(II) to Cr(III) while replacing halogens). In a niche exploit, certain tricarbonyl(arene)chromium complexes display benzylic activation.
Organochromium compounds
[edit]Organochromium compounds can be divided into these broad compound classes:
- Sandwich compounds: chromocene Cp2Cr and Bis(benzene)chromium derivatives (ArH)2Cr. More commonly studied are half-sandwich complexes such as (η6-C6H5OMe)Cr(CO)3.
- Chromium carbenes (R1)(R2)C::CrLn and carbynes (RC:::CrLn)
- Chromium(III) complexes RCrL5.[16]
- Complexes of chromium carbonyl anion and cation (e.g Na4Cr(CO)4).[17][18]
Ethylene polymerization and oligomerization
[edit]Chromium catalysts are important in ethylene polymerization.[19] The Phillips catalyst is prepared by impregnating chromium(VI) oxide on silica followed activation in dry air at high temperatures. The bright yellow catalyst becomes reduced by the ethylene to afford a probable Cr(II) species that is catalytically active.[20] A related catalytic systems developed by Union Carbide and DSM are also based on silica with chromocene and other chromium complexes. How these catalysts work is unclear. One model system describes it as coordination polymerization:
With two THF ligands the catalyst is stable but in dichloromethane one ligand is lost to form a 13 electron chromium intermediate. This enables side-on addition of an ethylene unit and a polymer chain can grow by migratory insertion.
Chromium compounds also catalyse the trimerization of ethylene to produce the monomer 1-hexene.[21][22]
References
[edit]- ^ Review: Carbon-Carbon Bond Formations Involving Organochromium(III) Reagents Furstner, A. Chem. Rev.; (Review); 1999; 99(4); 991-1046. doi:10.1021/cr9703360
- ^ Review: From Hein to Hexene: Recent Advances in the Chemistry of Organochromium -Complexes Jolly, P. W. Acc. Chem. Res.; (Article); 1996; 29(11); 544-551. doi:10.1021/ar9502588
- ^ Hein, F. (1919). "Notiz über Chromorganoverbindungen". Berichte der Deutschen Chemischen Gesellschaft (A and B Series). 52: 195–196. doi:10.1002/cber.19190520126.
- ^ Zeiss, Harold H.; Tsutsui, Minoru (1957). "π-Complexes of the Transition Metals. I. Hein's Polyaromatic Chromium Compounds". J. Am. Chem. Soc. 79 (12): 3062–3066. doi:10.1021/ja01569a019.
- ^ Fischer, E. Otto; Seus, Dietlinde (1956). "Zur Frage der Struktur der Chrom-phenyl-Verbindungen. Über Aromatenkomplexe von Metallen VI". Chemische Berichte. 89 (8): 1809–1815. doi:10.1002/cber.19560890803.
- ^ Hein, Fr. (1956). "Zur Frage der Struktur der Chrom-phenyl-Verbindungen. Bemerkungen zur Abhandlung von e. O. Fischer und D. Seus". Chemische Berichte. 89 (8): 1816–1821. doi:10.1002/cber.19560890804.
- ^ Fischer, E. O.; Hafner, W. Z. Naturforsch. 1953, 8b, 444.
- ^ Anet, F. A. L.; Leblanc, E. (1957). "A Novel Organo-Chromium Compound". Journal of the American Chemical Society. 79 (10): 2649–2650. doi:10.1021/ja01567a080.
- ^ Reduction of Organic Halides by Chromium(II). Mechanism of the Formation of Benzylchromium Ion Jay K. Kochi, Dennis D. Davis J. Am. Chem. Soc.; 1964; 86(23); 5264-5271. doi:10.1021/ja01077a044
- ^ Kochi, Jay K.; Singleton, David M. (1968). "Stereochemistry of reductive elimination by chromium(II) complexes". Journal of the American Chemical Society. 90 (6): 1582–1589. doi:10.1021/ja01008a032.
- ^ Wilke, G. Cyclooligomerisation von Butadien und Übergangsmetall--Komplexe Angewandte Chemie, 1963 Volume 75, pp. 10-20. doi:10.1002/ange.19630750104
- ^ Wilke, G.; Arbeiten Bogdanovič, Nach B.; Borner, P.; Breil, H.; Hardt, P.; Heimbach, P.; Herrmann, G.; Kaminsky, H.-J.; Keim, W.; Kröner, M.; Müller, Herbert; Müller, Ernst Willi; Oberkirch, W.; Schneider, J.; Stedefeder, J.; Tanaka, K.; Weyer, K.; Wilke, G. (1963). "Cyclooligomerisation von Butadien und Übergangsmetall-π-Komplexe". Angewandte Chemie. 75 (1): 10–20. Bibcode:1963AngCh..75...10W. doi:10.1002/ange.19630750104.
- ^ Karol, Frederick J.; Karapinka, George L.; Wu, Chisung; Dow, Alan W.; Johnson, Robert N.; Carrick, Wayne L. (1972). "Chromocene catalysts for ethylene polymerization: Scope of the polymerization". Journal of Polymer Science Part A-1: Polymer Chemistry. 10 (9): 2621–2637. Bibcode:1972JPoSA..10.2621K. doi:10.1002/pol.1972.150100910.
- ^ Fischer, Ernst Otto; Maasböl, Alfred (1967). "Übergangsmetall-Carben-Komplexe, II. Phenylmethoxycarben- und Methylmethoxycarben-pentacarbonyl-chrom, -molybdän, -wolfram und -cyclopentadienyl-dicarbonyl-mangan". Chemische Berichte. 100 (7): 2445–2456. doi:10.1002/cber.19671000744.
- ^ Fischer, Ernst Otto; Kreis, Gerhard; Kreiter, Cornelius G.; Müller, Jörn; Huttner, Gottfried; Lorenz, Hans (1973). "trans-Halogeno[alkyl(aryl)carbyne]tetracarbonyl Complexes of Chromium, Molybdenum, and Tungsten —A New Class of Compounds Having a Transition Metal-Carbon Triple Bond". Angewandte Chemie International Edition. 12 (14): 563. doi:10.1002/anie.197305641.
- ^ James H. Espenson Chemistry of Organochromium(III) Complexes Acc. Chem. Res.; 1992, volume 25, pp. 222-227. doi:10.1021/ar00017a003
- ^ Herrmann, Wolfgang A. (2014-05-14). Synthetic Methods of Organometallic and Inorganic Chemistry, Volume 7, 1997: Volume 7: Transition Metals Part 1. Georg Thieme Verlag. ISBN 978-3-13-179231-0.
- ^ Bohnenberger, Jan; Feuerstein, Wolfram; Himmel, Daniel; Daub, Michael; Breher, Frank; Krossing, Ingo (2019-02-07). "Stable salts of the hexacarbonyl chromium(I) cation and its pentacarbonyl-nitrosyl chromium(I) analogue". Nature Communications. 10 (1): 624. Bibcode:2019NatCo..10..624B. doi:10.1038/s41467-019-08517-2. ISSN 2041-1723. PMC 6367395. PMID 30733449.
- ^ Klaus H. Theopold Organochromium(III) chemistry: a neglected oxidation state Acc. Chem. Res. 1990, volume 23, pp. 263-270. doi:10.1021/ar00176a005
- ^ Kenneth S. Whiteley; T. Geoffrey Heggs; Hartmut Koch; Ralph L. Mawer; Wolfgang Immel (2005). "Polyolefins". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_487. ISBN 3-527-30673-0.
- ^ John T. Dixon, Mike J. Green, Fiona M. Hess, David H. Morgan "Advances in selective ethylene trimerisation – a critical overview" Journal of Organometallic Chemistry 2004, Volume 689, Pages 3641-3668. doi:10.1016/j.jorganchem.2004.06.008
- ^ Agapie, Theodor (2011). "Selective Ethylene Oligomerization: Recent Advances in Chromium Catalysis and Mechanistic Investigations". Coordination Chemistry Reviews. 255 (7–8): 861–880. doi:10.1016/j.ccr.2010.11.035.


 French
French Deutsch
Deutsch