芳基环己胺 - 维基百科,自由的百科全书
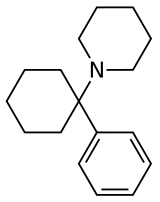
芳基环己胺衍生物(英語:Arylcyclohexylamine、arylcyclohexamines或arylcyclohexanamines)是一类含氮有机化合物,常用作药品和狡詐家藥物和试验药物,其特点是环己烷的一个碳原子上连接了氨基的N原子和芳香基,常见的有乙环利定、苯环己哌啶等[1]。


化合物
[编辑]| 结构 | 化合物 | 分子式 | 芳香基团 | 含N取代基 | 环己烷侧取代基 | CAS编号 |
|---|---|---|---|---|---|---|
 | PCA[2] | C12H17N | 苯基 | NH2 | - | 1934-71-0 |
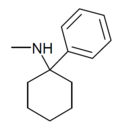 | PCM[2] | C13H19N | 苯基 | 甲氨基 | - | 2201-16-3 |
 | 乙环利定(PCE) | C14H21N | 苯基 | 乙氨基 | - | 2201-15-2 |
 | PCPr[3] | C15H23N | 苯基 | 正丙氨基 | - | 18949-81-0 |
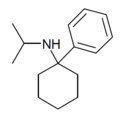 | PCiP | C15H23N | 苯基 | 异丙氨基 | - | 1195-42-2 |
 | PCAL [4] | C15H21N | 苯基 | 烯丙氨基 | - | 2185-95-7 |
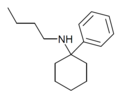 | PCBu | C16H25N | 苯基 | 正丁氨基 | - | 73166-29-7 |
 | PCEOH | C14H21NO | 苯基 | 羟乙氨基 | - | 2201-22-1 |
 | PCMEA[5] | C15H23NO | 苯基 | 甲氧乙氨基 | - | 2201-57-2 |
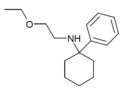 | PCEEA | C16H25NO | 苯基 | 乙氧乙氨基 | - | 1072895-05-6 |
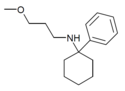 | PCMPA | C16H25NO | 苯基 | 甲氧丙氨基 | - | 2201-58-3 |
 | PCDM[2] | C14H21N | 苯基 | 二甲氨基 | - | 2201-17-4 |
 | 二乙环利定(PCDE) | C16H25N | 苯基 | 二乙氨基 | - | 2201-19-6 |
 | 2-HO-PCP[6] | C17H25NO | 苯基 | 哌啶基 | 2-羟基 | 94852-58-1 |
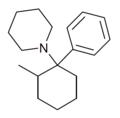 | 2-Me-PCP[7] | C18H27N | 苯基 | 哌啶基 | 2-甲基 | 59397-29-4 |
 | 2-MeO-PCP[8] | C18H27NO | 苯基 | 哌啶基 | 2-甲氧基 | 78636-34-7 |
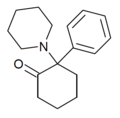 | 2-Keto-PCP | C17H23NO | 苯基 | 哌啶基 | 2-酮 | 101688-16-8 |
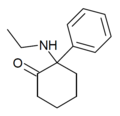 | 2-氧代-PCE("O-PCE") | C14H19NO | 苯基 | 乙氨基 | 2-酮 | 6740-82-5 |
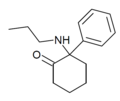 | 2-Keto-PCPr | C15H21NO | 苯基 | 正丙氨基 | 2-酮 | |
 | 4-Methyl-PCP | C18H27N | 苯基 | 哌啶基 | 4-甲基 | 19420-52-1 |
 | 4-Keto-PCP[9] | C17H23NO | 苯基 | 哌啶基 | 4-酮 | 65620-13-5 |
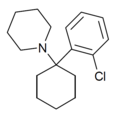 | 2'-Cl-PCP | C17H24ClN | 邻氯苯基 | 哌啶基 | - | 2201-31-2 |
 | 3'-Cl-PCP | C17H24ClN | 间氯苯基 | 哌啶基 | - | 2201-32-3 |
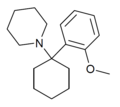 | 2'-MeO-PCP | C18H27NO | 邻甲氧基苯基 | 哌啶基 | - | 2201-34-5 |
 | 3'-F-PCP[10] | C17H24FN | 间氟苯基 | 哌啶基 | - | 89156-99-0 |
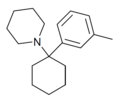 | 3-甲基苯环己哌啶(3'-Me-PCP)[11] | C18H27N | 间甲苯基 | 哌啶基 | - | 2201-30-1 |
 | 3'-Me-PCPy | C17H25N | 间甲苯基 | 吡咯基 | - | 1622348-63-3 |
 | 3'-NH2-PCP | C17H26N2 | 间氨基苯基 | 哌啶基 | - | 72242-00-3 |
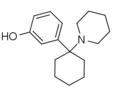 | 3-羥基苯環己哌啶(3'-HO-PCP) | C17H25NO | 间羟基苯基 | 哌啶基 | - | 79787-43-2 |
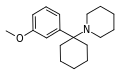 | 3-甲氧基苯環己哌啶(3'-MeO-PCP) | C18H27NO | 间甲氧基苯基 | 哌啶基 | - | 72242-03-6 |
 | 3',4'-MD-PCP | C18H25NO2 | 3,4-亚甲二氧基苯基 | 哌啶基 | - | |
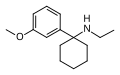 | 3'-MeO-PCE | C15H23NO | 间甲氧基苯基 | 乙氨基 | - | 1364933-80-1 |
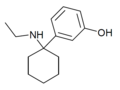 | 3'-HO-PCE | 间羟基苯基 | 乙氨基 | - | ||
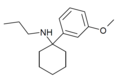 | 3'-MeO-PCPr | 间甲氧基苯基 | 正丙氨基 | - | 1364933-81-2 | |
 | 3'-HO-PCPr | 间羟基苯基 | 正丙氨基 | - | ||
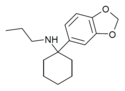 | 3',4'-MD-PCPr | 3,4-亚甲二氧基苯基 | 正丙氨基 | - | ||
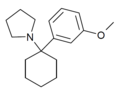 | 3'-MeO-PCPy[11] | 间甲氧基苯基 | 吡咯基 | - | 1364933-79-8 | |
 | 4-羥基苯環己哌啶(4'-HO-PCP) | C17H25NO | 对羟基苯基 | 哌啶基 | - | 66568-88-5 |
 | 4-甲氧基苯環己哌啶(4'-MeO-PCP) | C18H27NO | 对甲氧基苯基 | 哌啶基 | - | 2201-35-6 |
 | 4'-MeO-PCE | 对甲氧基苯基 | 乙氨基 | - | ||
 | 4'-F-PCP[10] | p-Fluorophenyl | 哌啶基 | - | 22904-99-0 | |
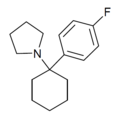 | 4'-F-PCPy | p-Fluorophenyl | 吡咯基 | - | ||
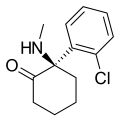 | Arketamine | C13H16ClNO | 邻氯苯基 | 甲氨基 | 2-酮 | 33643-49-1 |
 | Deschloroketamine | C13H17NO | 苯基 | 甲氨基 | 2-酮 | 7063-30-1 |
 | Esketamine | C13H16ClNO | 邻氯苯基 | 甲氨基 | 2-酮 | 33643-46-8 |
 | 氯胺酮 | C13H16ClNO | 邻氯苯基 | 甲氨基 | 2-酮 | 6740-88-1 |
 | Hydroxynorketamine | C12H14ClNO2 | 邻氯苯基 | NH2 | 2-酮,6-羟基 | 81395-70-2 |
 | Ethketamine | C14H18ClNO | 邻氯苯基 | 乙氨基 | 2-酮 | 1354634-10-8 |
 | NPNK | C15H20ClNO | 邻氯苯基 | 正丙氨基 | 2-酮 | 2749326-65-4 |
 | Methoxyketamine | C14H19NO2 | 邻甲氧基苯基 | 甲氨基 | 2-酮 | 7063-51-6 |
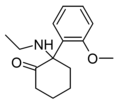 | 2-MeO-NEK[12] | C15H21NO2 | 邻甲氧基苯基 | 乙氨基 | 2-酮 | |
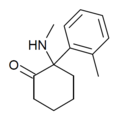 | oMDCK[13] | C14H19NO | 邻甲苯基 | 甲氨基 | 2-酮 | 7063-37-8 |
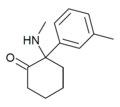 | mMDCK | C14H19NO | 间甲苯基 | 甲氨基 | 2-酮 | |
 | meta-Ketamine | C13H16ClNO | 间氯苯基 | 甲氨基 | 2-酮 | 7063-53-8 |
 | iso-Ketamine | C13H16ClNO | 邻氯苯基 | 甲氨基 | 4-酮 | |
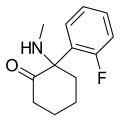 | 氟胺酮 | C13H16FNO | 邻氟苯基 | 甲氨基 | 2-酮 | 111982-50-4 |
 | 3-氟脱氯氯胺酮 | C13H16FNO | 间氟苯基 | 甲氨基 | 2-酮 | 2657761-23-2 |
 | Bromoketamine | 邻溴苯基 | 甲氨基 | 2-酮 | 120807-70-7 | |
 | TFMDCK | o-Trifluoromethylphenyl | 甲氨基 | 2-酮 | 1782149-73-8 | |
 | SN 35210[14] | 邻氯苯基 | Carbomethoxybutylamino | 2-酮 | 1450615-41-4 | |
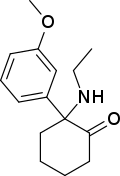 | Methoxetamine | C15H21NO2 | 间甲氧基苯基 | 乙氨基 | 2-酮 | 1239943-76-0 |
 | Methoxmetamine | C14H19NO2 | 间甲氧基苯基 | 甲氨基 | 2-酮 | 1781829-56-8 |
 | Methoxpropamine | C16H23NO2 | 间甲氧基苯基 | 正丙氨基 | 2-酮 | 2504100-71-2 |
 | MXiPr | C16H23NO2 | 间甲氧基苯基 | 异丙氨基 | 2-酮 | |
 | Ethoxetamine | C16H23NO2 | 间乙氧基苯基 | 乙氨基 | 2-酮 | |
 | Deoxymethoxetamine (3-Me-2'-Oxo-PCE) | 间甲苯基 | 乙氨基 | 2-酮 | 2666932-45-0 | |
 | Br-MXE | 2-溴-5-甲氧基苯基 | 乙氨基 | 2-酮 | ||
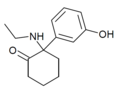 | Hydroxetamine (HXE) | 间羟基苯基 | 乙氨基 | 2-酮 | 1620054-73-0 | |
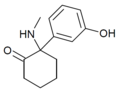 | HXM | 间羟基苯基 | 甲氨基 | 2-酮 | ||
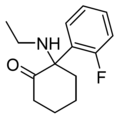 | 2F-NENDCK | C14H18FNO | 邻氟苯基 | 乙氨基 | 2-酮 | |
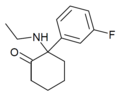 | Fluorexetamine (FXE) | C14H18FNO | 间氟苯基 | 乙氨基 | 2-酮 | |
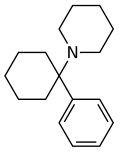 | 苯环己哌啶(PCP) | C17H25N | 苯基 | 哌啶基 | - | 77-10-1 |
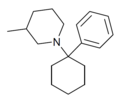 | PC3MP | C18H27N | 苯基 | 3-甲基哌啶基 | - | 2201-41-4 |
 | PC4MP | C18H27N | 苯基 | 4-甲基哌啶基 | - | 2201-42-5 |
 | 咯环利定(PCPy) | C16H23N | 苯基 | 吡咯基 | - | 2201-39-0 |
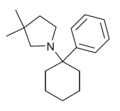 | PCDMPy | C18H27N | 苯基 | 3,3-二甲基吡咯基 | - | |
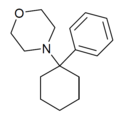 | PCMo | C16H23NO | 苯基 | 吗啉基 | - | 2201-40-3 |
 | Methoxy-PCM[15] (2'-MeO-PCMo) | 邻甲氧基苯基 | Morpholine | - | 1314323-88-0 | |
 | 3'-MeO-PCMo | 间甲氧基苯基 | Morpholine | - | 138873-80-0 | |
 | 4'-MeO-PCMo | 对间甲氧基苯基 | Morpholine | - | ||
 | Methyl-PCM[16] (4'-Me-PCMo) | 对甲苯基 | Morpholine | - | 120803-52-3 | |
 | Hydroxy-methyl-PCM | 2-Methyl-4-hydroxyphenyl | Morpholine | - | 1314323-89-1 | |
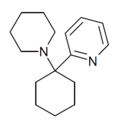 | PYCP [17] | 2-Pyridinyl | 哌啶基 | - | ||
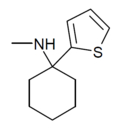 | TCM | 2-噻吩基 | 甲氨基 | - | 139401-07-3 | |
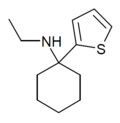 | TCE | 2-噻吩基 | 乙氨基 | - | 101589-62-2 | |
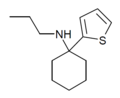 | TCPr [18] | 2-噻吩基 | 正丙氨基 | - | ||
 | Tenocyclidine (TCP) | 2-噻吩基 | 哌啶基 | - | 21500-98-1 | |
 | T3CP | 3-噻吩基 | 哌啶基 | - | 19420-50-9 | |
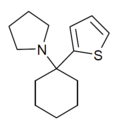 | TCPy | 2-噻吩基 | 吡咯基 | - | 22912-13-6 | |
 | Tiletamine | 2-噻吩基 | 乙氨基 | 2-酮 | 14176-49-9 | |
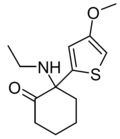 | MXTE | 4-Methoxy-2-thienyl | 乙氨基 | 2-酮 | ||
 | Gacyclidine | 2-噻吩基 | 哌啶基 | 2-Methyl | 68134-81-6 | |
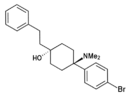 | BDPC | p-Bromophenyl | Dimethylamino | 4-Phenethyl-4-hydroxy | 77239-98-6 | |
 | C-8813 | p-Bromophenyl | Dimethylamino | 4-(thiophen-2-yl)ethyl-4-hydroxy | 616898-54-5 | |
 | Dimetamine[19] | 对甲苯基 | Dimethylamino | 4-酮 | 65619-06-9 | |
 | 3''-OH-2'-Me-PCP [20] | C18H27NO | 邻甲苯基 | 3-Hydroxypiperidine | - | |
 | 4''-Ph-4''-OH-PCP [21] | 苯基 | 4-Phenyl-4-hydroxypiperidine | - | 77179-39-6 | |
 | BTCP[22] | Benzothiophen-2-yl | 哌啶基 | - | 112726-66-6 | |
 | BTCPy[23] | Benzothiophen-2-yl | 吡咯基 | - | ||
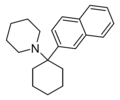 | GK-189[24] | Naphthalen-2-yl | 哌啶基 | - | 81490-58-6 |
参考文献
[编辑]- ^ 4-(1-phenyl-cyclohexyl)-morpholine. CAS Number Search - chemsrc.com. chemsrc. [15 March 2021].
- ^ 2.0 2.1 2.2 Thurkauf A, de Costa B, Yamaguchi S, Mattson MV, Jacobson AE, Rice KC, Rogawski MA. Synthesis and anticonvulsant activity of 1-phenylcyclohexylamine analogues. Journal of Medicinal Chemistry. May 1990, 33 (5): 1452–8. PMID 2329567. doi:10.1021/jm00167a027.
- ^ Sauer C, Peters FT, Staack RF, Fritschi G, Maurer HH. Metabolism and toxicological detection of a new designer drug, N-(1-phenylcyclohexyl)propanamine, in rat urine using gas chromatography-mass spectrometry. Journal of Chromatography A. April 2008, 1186 (1–2): 380–90. PMID 18035363. doi:10.1016/j.chroma.2007.11.002.
- ^ Kalir A, Teomy S, Amir A, Fuchs P, Lee SA, Holsztynska EJ, et al. N-allyl analogues of phencyclidine: chemical synthesis and pharmacological properties. Journal of Medicinal Chemistry. October 1984, 27 (10): 1267–71. PMID 6481761. doi:10.1021/jm00376a006.
- ^ Sauer C, Peters FT, Schwaninger AE, Meyer MR, Maurer HH. Investigations on the cytochrome P450 (CYP) isoenzymes involved in the metabolism of the designer drugs N-(1-phenyl cyclohexyl)-2-ethoxyethanamine and N-(1-phenylcyclohexyl)-2-methoxyethanamine. Biochemical Pharmacology. February 2009, 77 (3): 444–50. PMID 19022226. doi:10.1016/j.bcp.2008.10.024.
- ^ 引用错误:没有为名为
pmid16229117的参考文献提供内容 - ^ Iorio MA, Tomassini L, Mattson MV, George C, Jacobson AE. Synthesis, stereochemistry, and biological activity of the 1-(1-phenyl-2-methylcyclohexyl)piperidines and the 1-(1-phenyl-4-methylcyclohexyl)piperidines. Absolute configuration of the potent trans-(-)-1-(1-phenyl-2-methylcyclohexyl)piperidine. Journal of Medicinal Chemistry. August 1991, 34 (8): 2615–23. PMID 1875352. doi:10.1021/jm00112a041.
- ^ Ahmadi A, Mahmoudi A. Synthesis with improved yield and study on the analgesic effect of 2-methoxyphencyclidine. Arzneimittel-Forschung. 2006, 56 (5): 346–50. PMID 16821645. S2CID 10370245. doi:10.1055/s-0031-1296732.
- ^ Ortiz DM, Custodio RJ, Abiero A, Botanas CJ, Sayson LV, Kim M, et al. The dopaminergic alterations induced by 4-F-PCP and 4-Keto-PCP may enhance their drug-induced rewarding and reinforcing effects: Implications for abuse. Addiction Biology. July 2021, 26 (4): e12981. PMID 33135332. S2CID 226234538. doi:10.1111/adb.12981.
- ^ 10.0 10.1 Ogunbadeniyi AM, Adejare A. Syntheses of fluorinated phencyclidine analogs. Journal of Fluorine Chemistry. 2002, 114: 39–42. doi:10.1016/S0022-1139(01)00565-6.
- ^ 11.0 11.1 Wallach J, De Paoli G, Adejare A, Brandt SD. Preparation and analytical characterization of 1-(1-phenylcyclohexyl)piperidine (PCP) and 1-(1-phenylcyclohexyl)pyrrolidine (PCPy) analogues. Drug Testing and Analysis. 2013, 6 (7–8): 633–50. PMID 23554350. doi:10.1002/dta.1468.
- ^ Sayson LV, Botanas CJ, Custodio RJ, Abiero A, Kim M, Lee HJ, et al. The novel methoxetamine analogs N-ethylnorketamine hydrochloride (NENK), 2-MeO-N-ethylketamine hydrochloride (2-MeO-NEK), and 4-MeO-N-ethylketamine hydrochloride (4-MeO-NEK) elicit rapid antidepressant effects via activation of AMPA and 5-HT2 receptors. Psychopharmacology. July 2019, 236 (7): 2201–2210. PMID 30891619. S2CID 83463722. doi:10.1007/s00213-019-05219-x.
- ^ WO 2021134086,Kruegel AC, Sames D, Hashimoto K,「Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disorders」,发表于1 July 2021,指定于Gilgamesh Pharmaceuticals, Inc.和The Trustees Of Columbia University In The City Of New York. (页面存档备份,存于互联网档案馆)
- ^ Harvey M, Sleigh J, Voss L, Pruijn F, Jose J, Gamage S, Denny W. Determination of the Hypnotic Potency in Rats of the Novel Ketamine Ester Analogue SN 35210. Pharmacology. 2015, 96 (5–6): 226–32. PMID 26352278. S2CID 36017002. doi:10.1159/000439598.
- ^ 引用错误:没有为名为
pmid21215770的参考文献提供内容 - ^ Ahmadi A, Khalili M, Hajikhani R, Naserbakht M. Synthesis and determination of acute and chronic pain activities of 1-[1-(4-methylphenyl) (cyclohexyl)] morpholine as a new phencyclidine derivative in rats. Arzneimittel-Forschung. 2011, 61 (2): 92–7. PMID 21428243. S2CID 8094521. doi:10.1055/s-0031-1296173.
- ^ Zarantonello P, Bettini E, Paio A, Simoncelli C, Terreni S, Cardullo F. Novel analogues of ketamine and phencyclidine as NMDA receptor antagonists. Bioorganic & Medicinal Chemistry Letters. April 2011, 21 (7): 2059–63. PMID 21334205. doi:10.1016/j.bmcl.2011.02.009.
- ^ Wallach J, Colestock T, Cicali B, Elliott SP, Kavanagh PV, Adejare A, et al. Syntheses and analytical characterizations of N-alkyl-arylcyclohexylamines (PDF). Drug Testing and Analysis. August 2016, 8 (8): 801–15 [2024-02-23]. PMID 26360516. S2CID 1599386. doi:10.1002/dta.1861. (原始内容存档 (PDF)于2023-08-10).
- ^ Lednicer D, VonVoigtlander PF, Emmert DE. 4-Amino-4-arylcyclohexanones and their derivatives, a novel class of analgesics. 1. Modification of the aryl ring. Journal of Medicinal Chemistry. April 1980, 23 (4): 424–30. PMID 7381841. doi:10.1021/jm00178a014.
- ^ Ahmadi A, Solati J, Hajikhani R, Onagh M, Javadi M. Synthesis and analgesic effects of 1-[1-(2-methylphenyl)(cyclohexyl)]-3-piperidinol as a new derivative of phencyclidine in mice. Arzneimittel-Forschung. 2010, 60 (8): 492–6. PMID 20863005. S2CID 24803623. doi:10.1055/s-0031-1296317.
- ^ Itzhak Y, Kalir A, Weissman BA, Cohen S. New analgesic drugs derived from phencyclidine. Journal of Medicinal Chemistry. May 1981, 24 (5): 496–9. PMID 7241506. doi:10.1021/jm00137a004.
- ^ Vignon J, Pinet V, Cerruti C, Kamenka JM, Chicheportiche R. [3H]N-[1-(2-benzo(b)thiophenyl)cyclohexyl]piperidine ([3H]BTCP): a new phencyclidine analog selective for the dopamine uptake complex. European Journal of Pharmacology. April 1988, 148 (3): 427–36. PMID 3384005. doi:10.1016/0014-2999(88)90122-7.
- ^ 引用错误:没有为名为
pmid8098066的参考文献提供内容 - ^ Kamenka JM, et al. Substituted cyclic amines and pharmaceutical composition containing them. Patent US5248686, 28 September 1993. [2024-02-23]. (原始内容存档于2023-10-14).


 French
French Deutsch
Deutsch